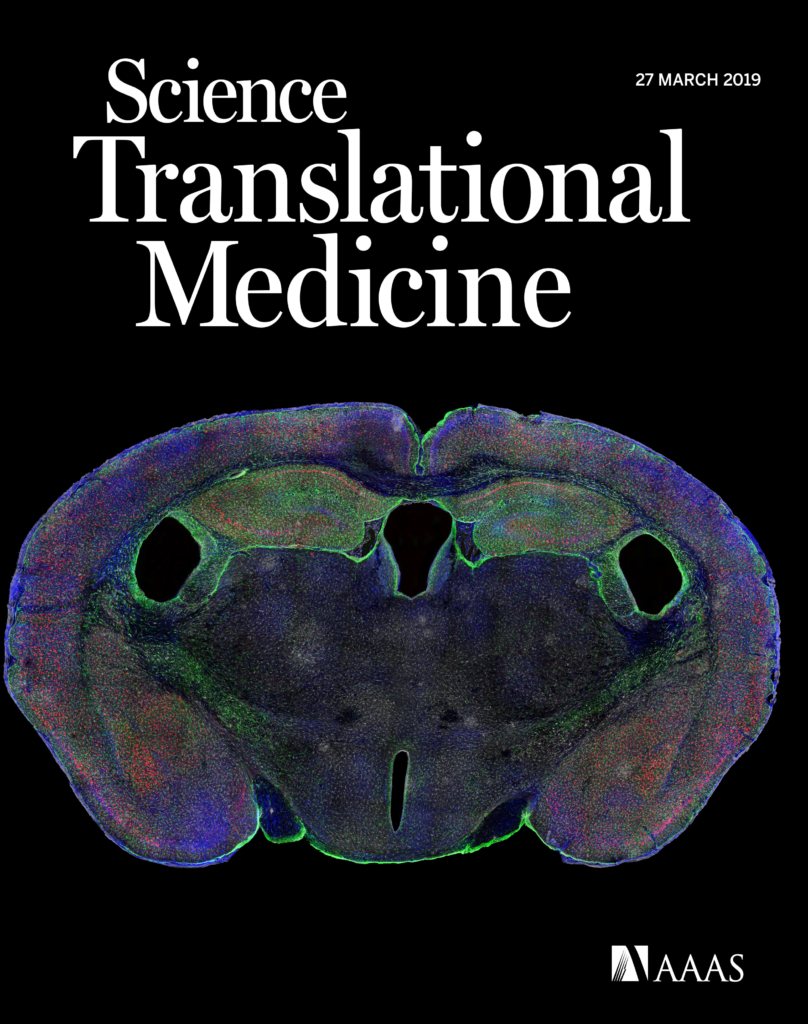
Our latest publication in the journal Science Translational Medicine is now avaliable! The article in its entirety can be found here. I was fortunate enough to provide the imaging data for this project as part of the Kosik Molecular and Cellular Neurobiology lab. I was also lucky enough to have my image selected as a the journal cover.
Authors: Israel Hernandez, myself, Michel Giroux, Celeste Karch, Daniel Boctor, Youssef Sibih, Nadia Storm, Antonio Dias, Cezary Zekanowski, Alex Kang, Cassidy Hinman, Vesna Cerovac, Elmer Guzman, Honjun Zhou, Alison Goate, Steve Fisher, Ana Cuervo, Ken Kosik.
Abstract:
Tau inclusions are a shared feature of many neurodegenerative diseases, among them frontotemporal dementia caused by tau mutations. Treatment approaches for these conditions include targeting posttranslational modifi-cations of tau proteins, maintaining a steady-state amount of tau, and preventing its tendency to aggregate. We discovered a new regulatory pathway for tau degradation that operates through the farnesylated protein, Rhes, a GTPase in the Ras family. Here, we show that treatment with the farnesyltransferase inhibitor lonafarnib reduced Rhes and decreased brain atrophy, tau inclusions, tau sumoylation, and tau ubiquitination in the rTg4510 mouse model of tauopathy. In addition, lonafarnib treatment attenuated behavioral abnormalities in rTg4510 mice and reduced microgliosis in mouse brain. Direct reduction of Rhes in the rTg4510 mouse by siRNA reproduced the results observed with lonafarnib treatment. The mechanism of lonafarnib action mediated by Rhes to reduce tau pathology was shown to operate through activation of lysosomes. We finally showed in mouse brain and in hu-man induced pluripotent stem cell–derived neurons a normal developmental increase in Rhes that was initially suppressed by tau mutations. The known safety of lonafarnib revealed in human clinical trials for cancer suggests that this drug could be repurposed for treating tauopathies.
News and Media Coverage: Scientific American, The Conversation, Gizmodo, UC Santa Barbara, San Francisco Chronicle.