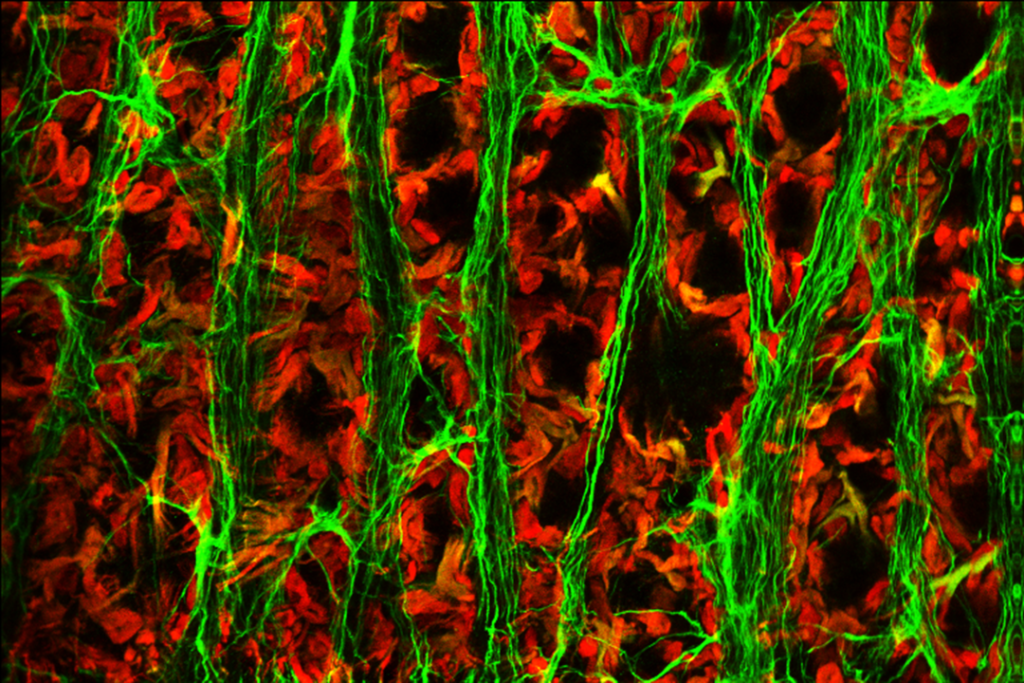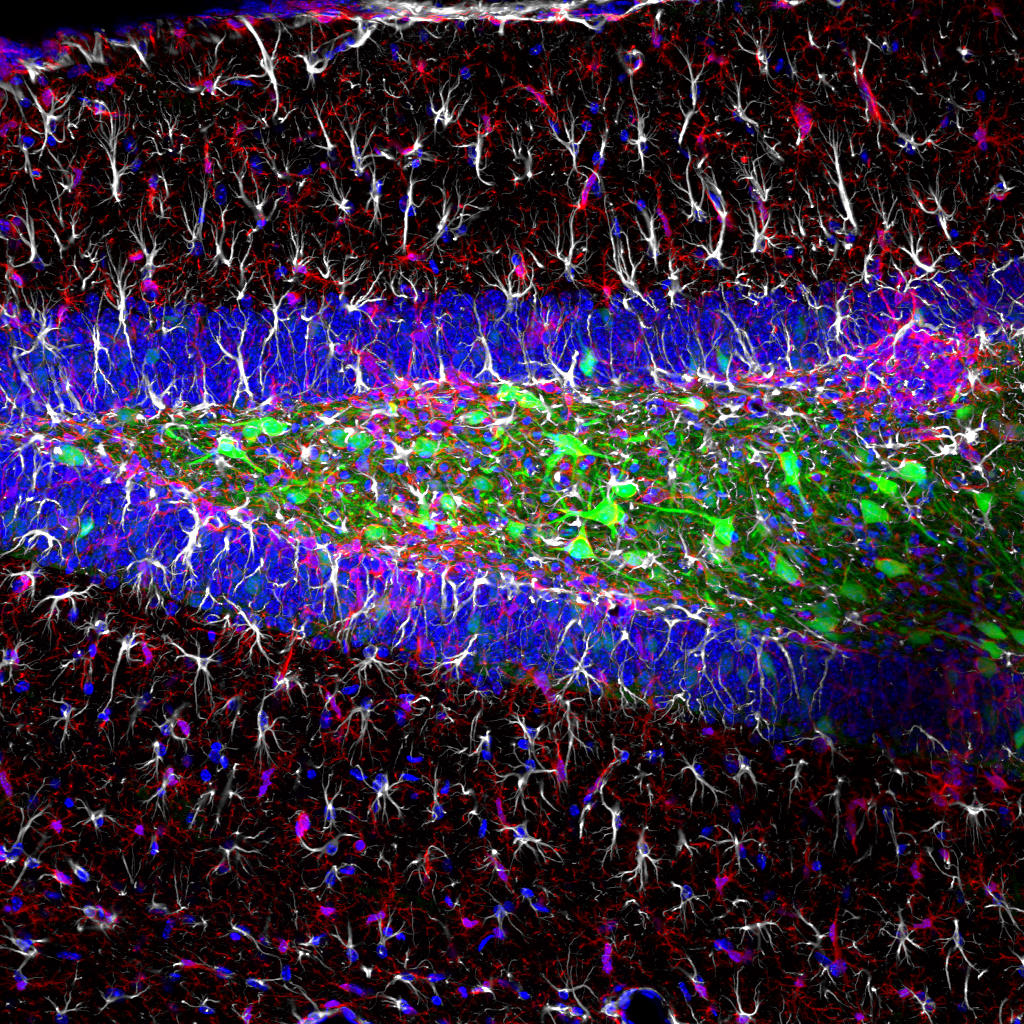
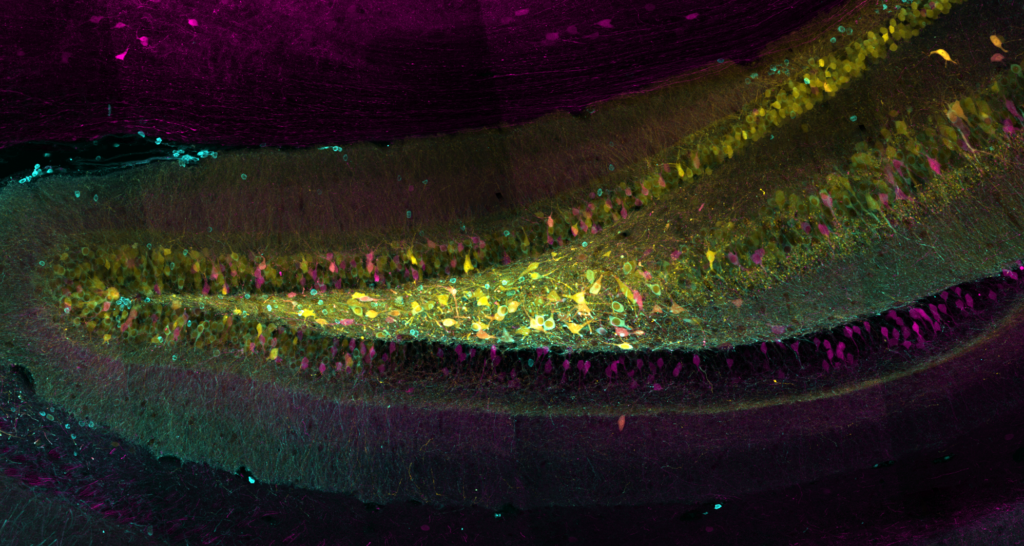
Our latest publication in the journal Nature is now avaliable!
Authors: Jennifer N. Rauch, Gabriel Luna, Elmer Guzman, Morgane Audouard, Collin Challis, Youssef E. Sibih, Carolina Leshuk, Israel Hernandez, Susanne Wegmann, Bradley T. Hyman, Viviana Gradniaru, Martin Kampmann, and Kenneth S. Kosik
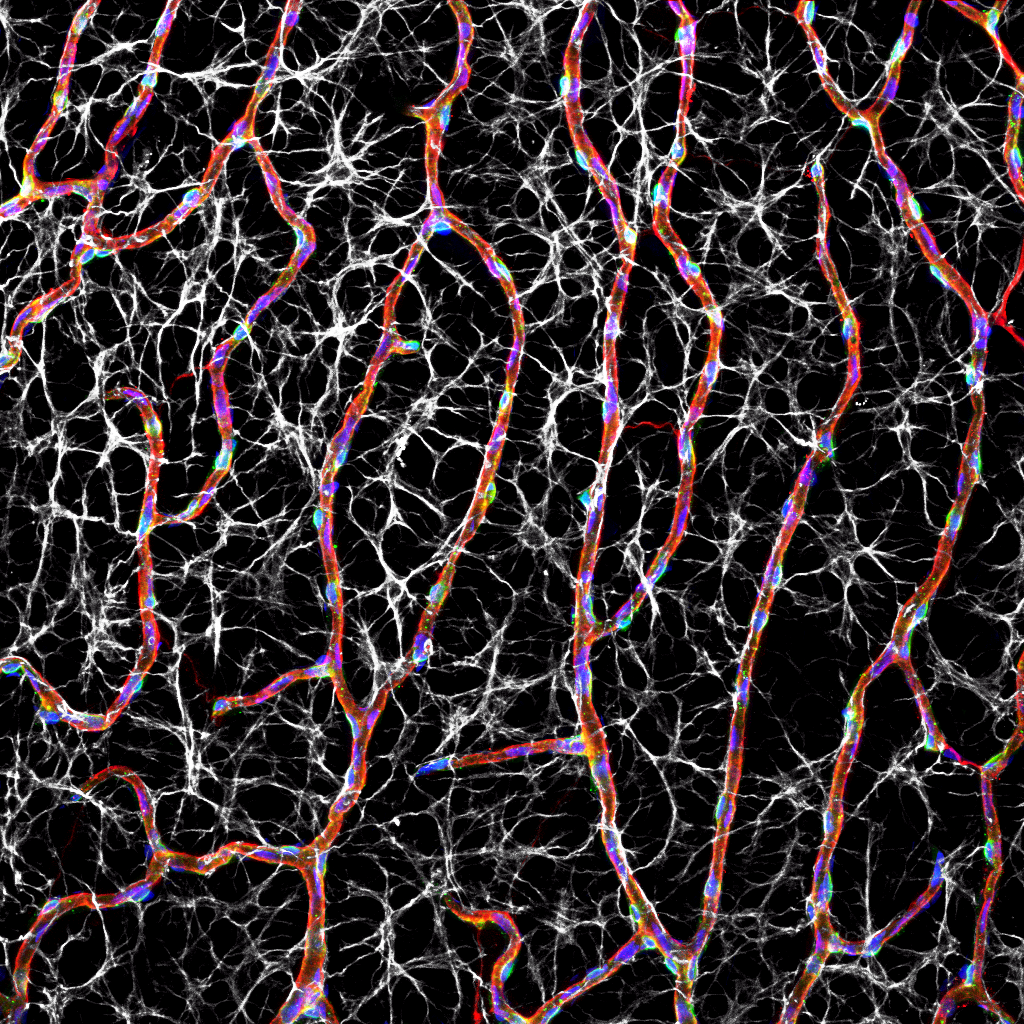
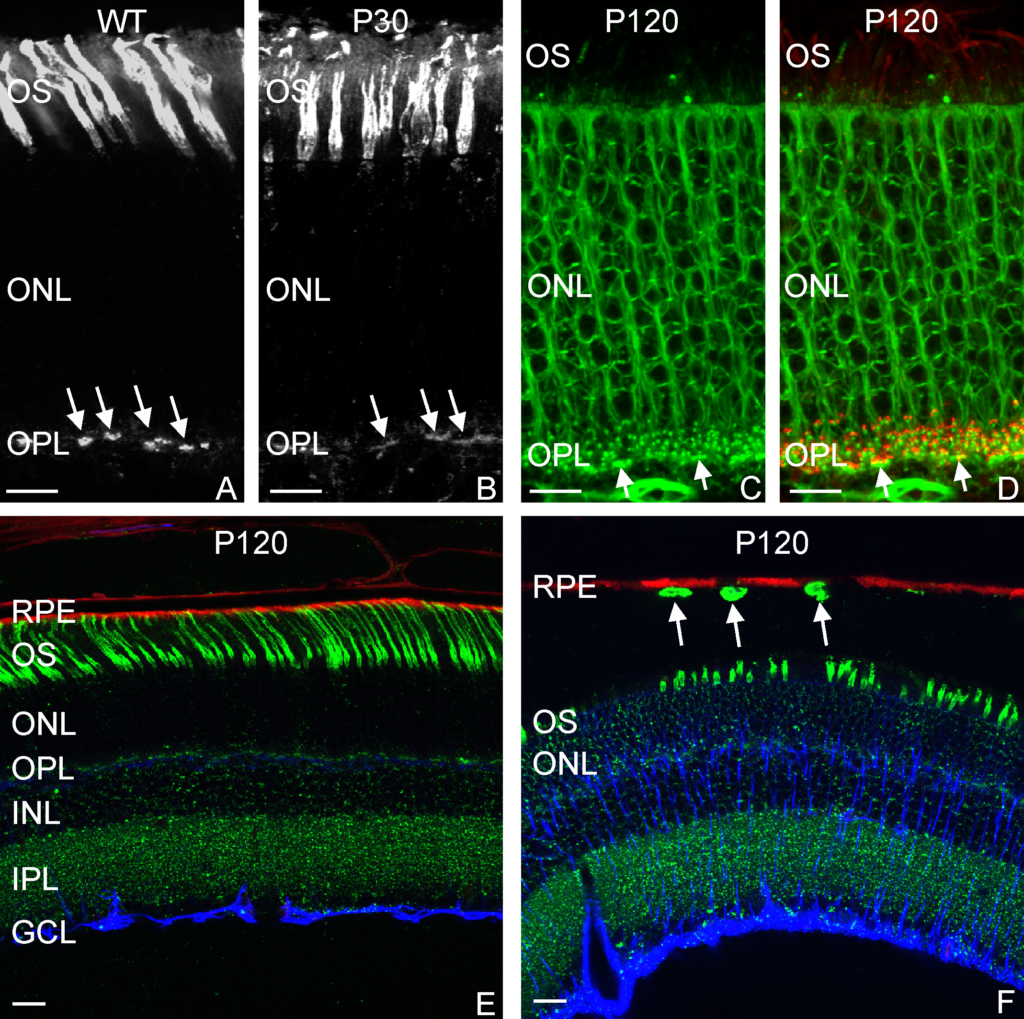
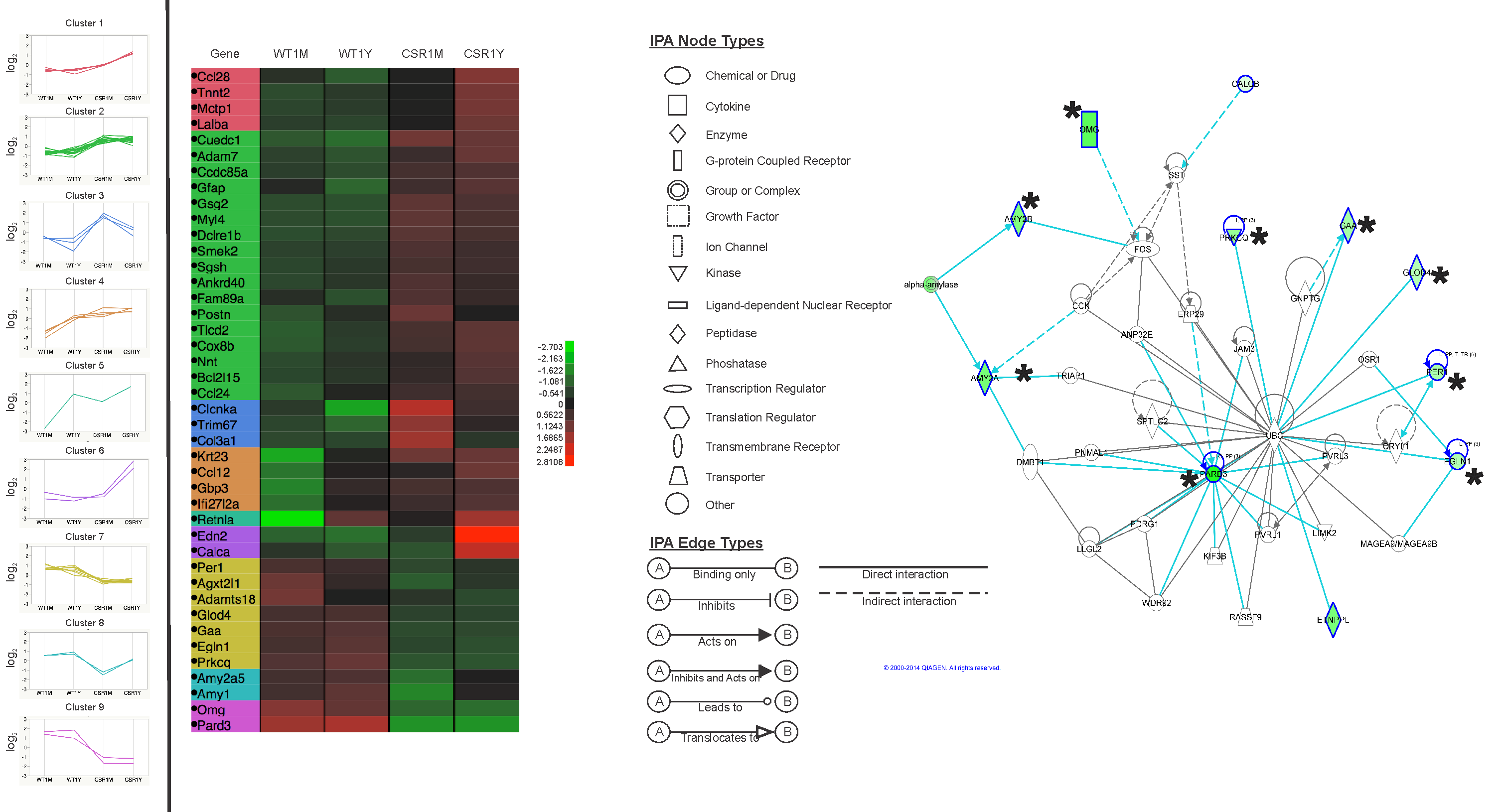
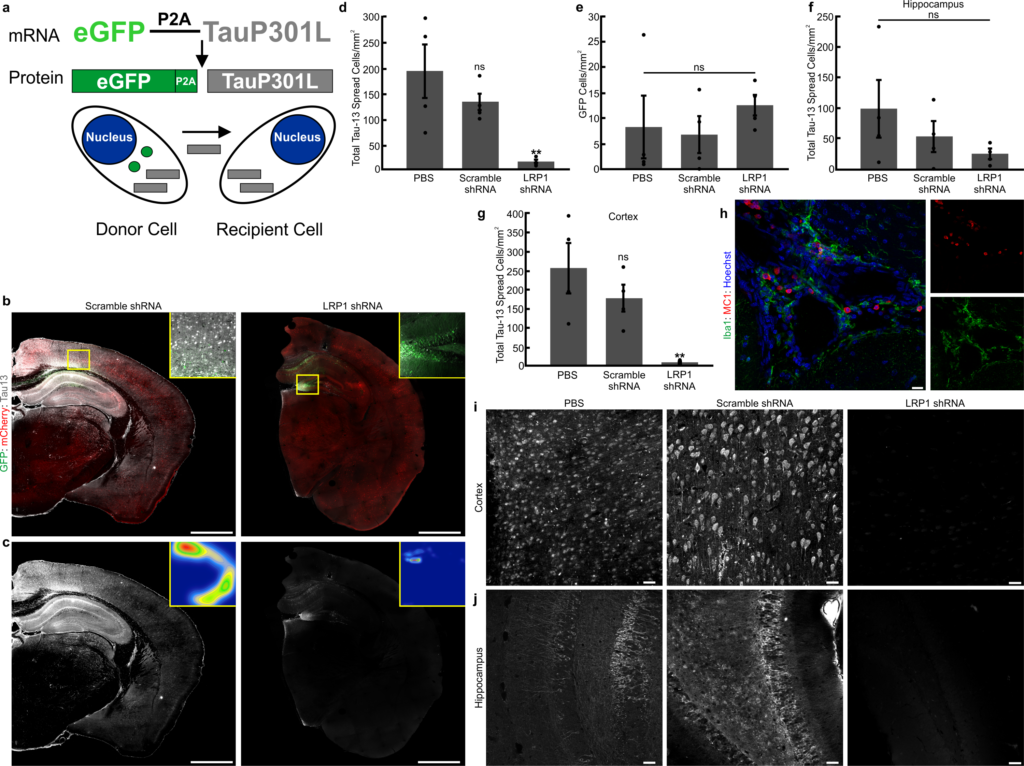
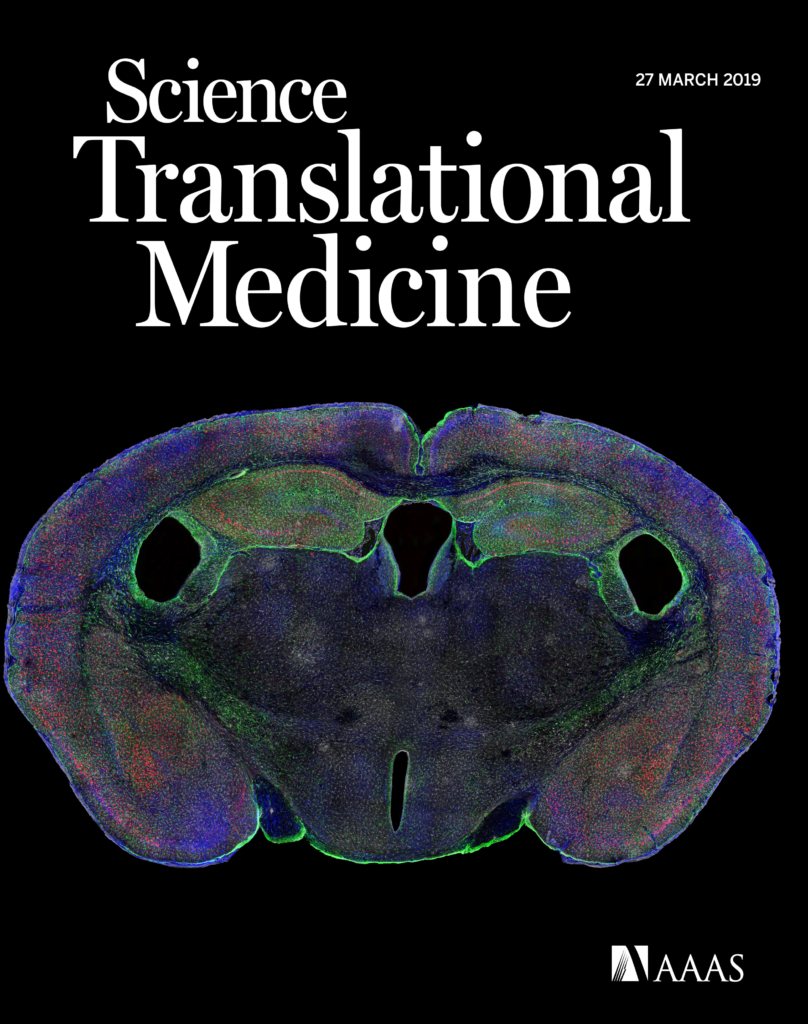
Abstract: The spread of protein aggregates during disease progression is a common theme underlying many neurodegenerative diseases. The microtubule-associated protein tau has a central role in the pathogenesis of several forms of dementia known as tauopathies—including Alzheimer’s disease, frontotemporal dementia and chronic traumatic encephalopathy. Progression of these diseases is characterized by the sequential spread and deposition of protein aggregates in a predictable pattern that correlates with clinical severity. This observation and complementary experimental studies have suggested that tau can spread in a prion-like manner, by passing to naive cells in which it templates misfolding and aggregation. However, although the propagation of tau has been extensively studied, the underlying cellular mechanisms remain poorly understood. Here we show that the low-density lipoprotein receptor-related protein 1 (LRP1) controls the endocytosis of tau and its subsequent spread. Knockdown of LRP1 significantly reduced tau uptake in H4 neuroglioma cells and in induced pluripotent stem cell-derived neurons. The interaction between tau and LRP1 is mediated by lysine residues in the microtubule-binding repeat region of tau. Furthermore, downregulation of LRP1 in an in vivo mouse model of tau spread was found to effectively reduce the propagation of tau between neurons. Our results identify LRP1 as a key regulator of tau spread in the brain, and therefore a potential target for the treatment of diseases that involve tau spread and aggregation.
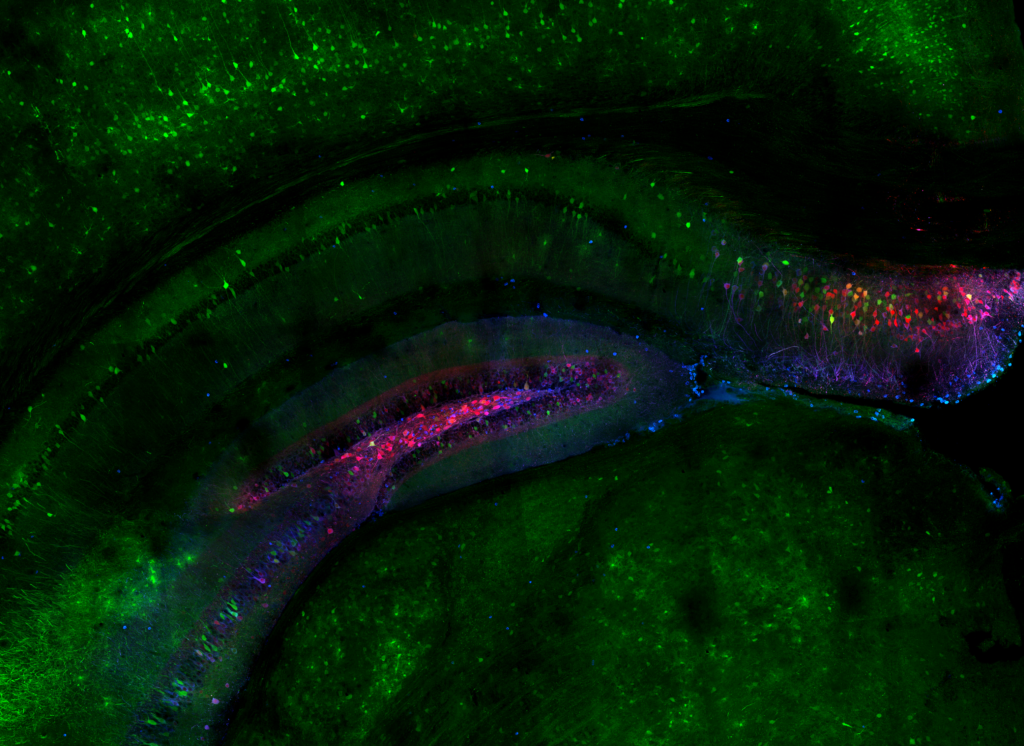
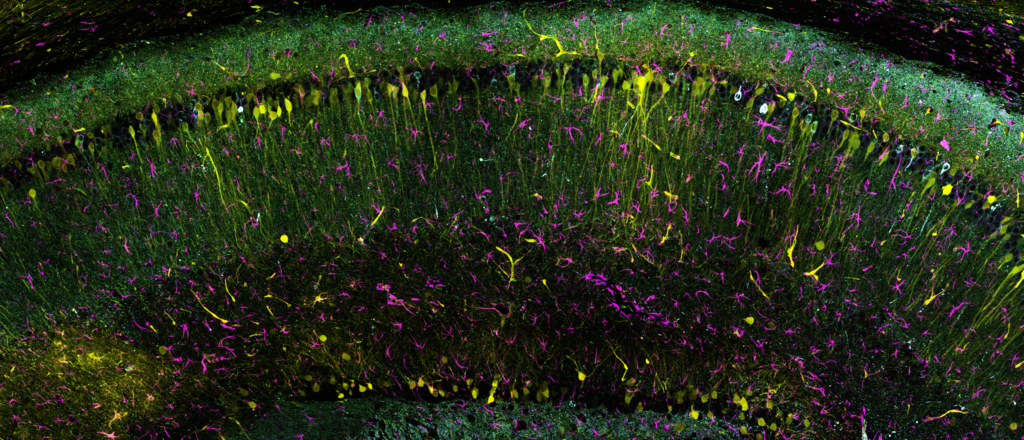
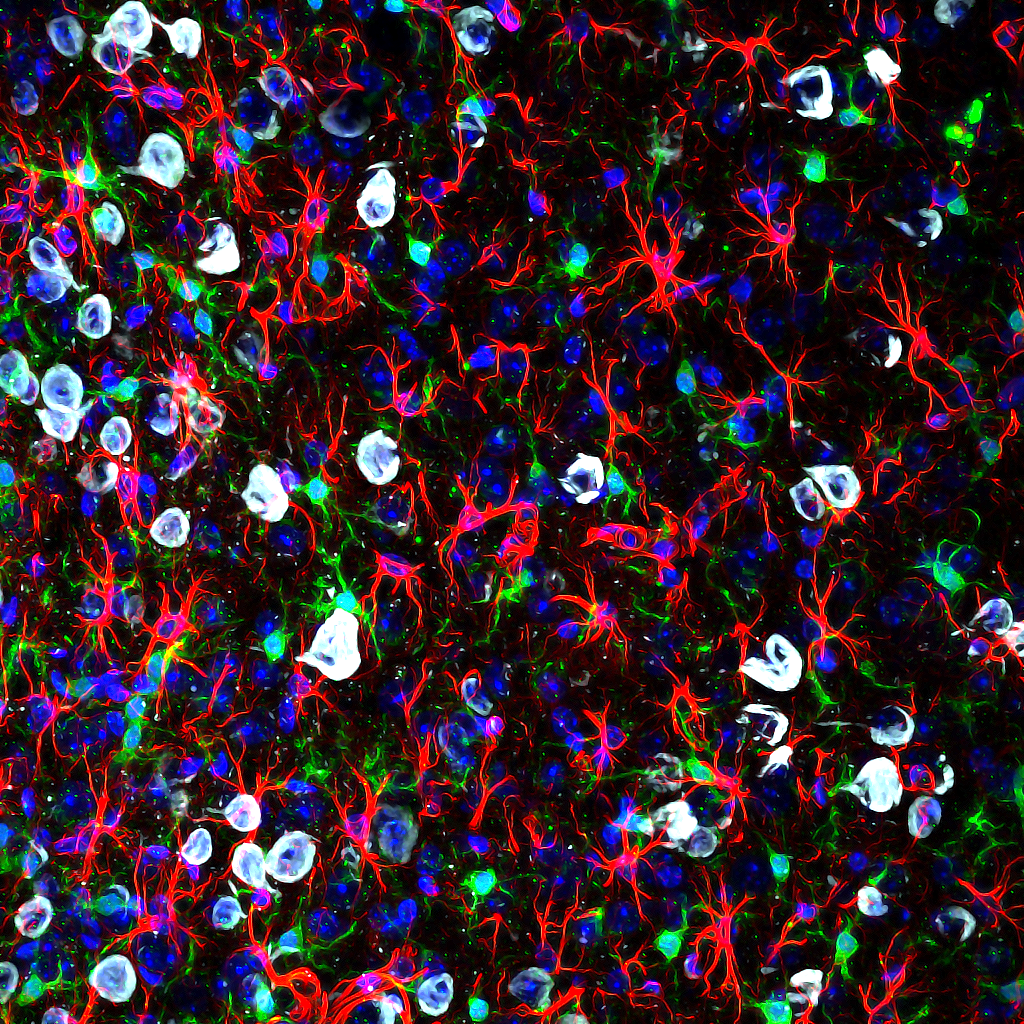
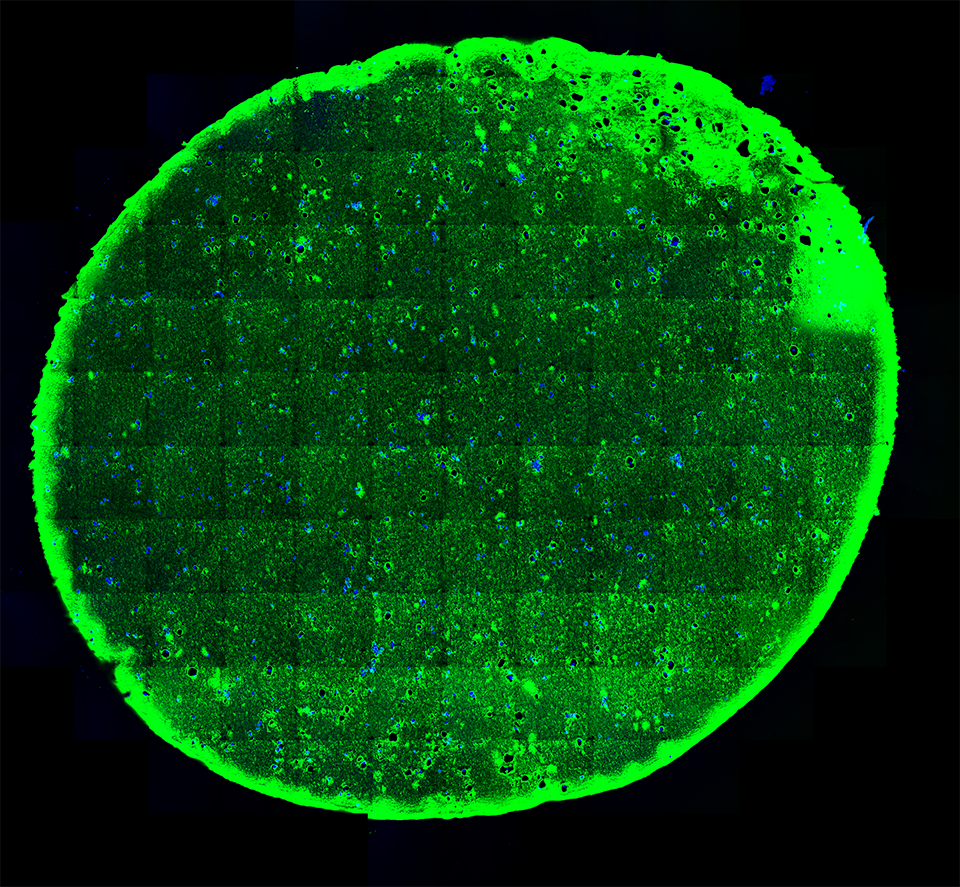
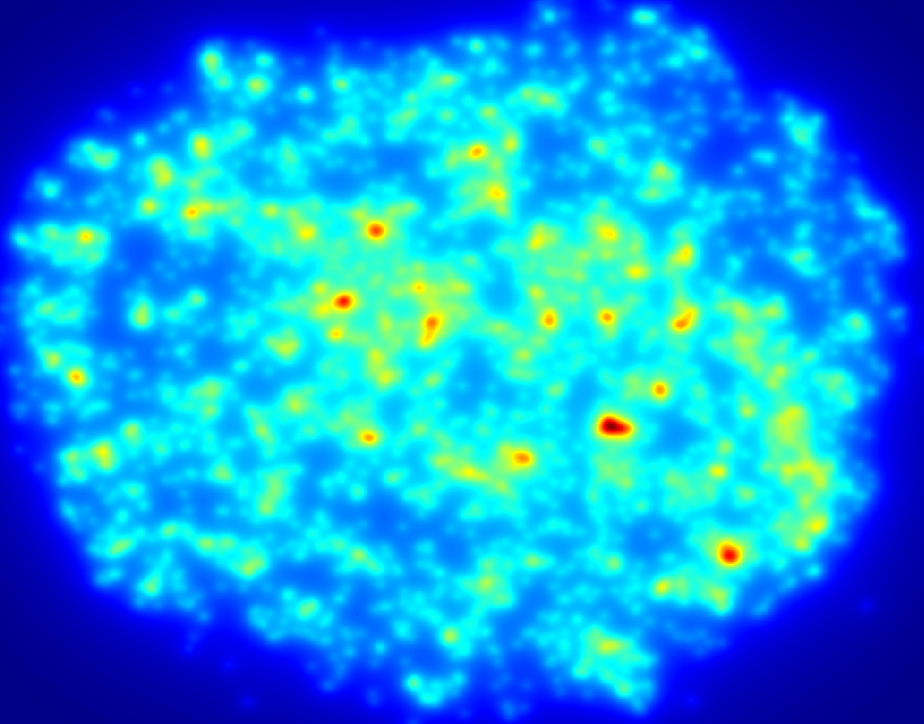
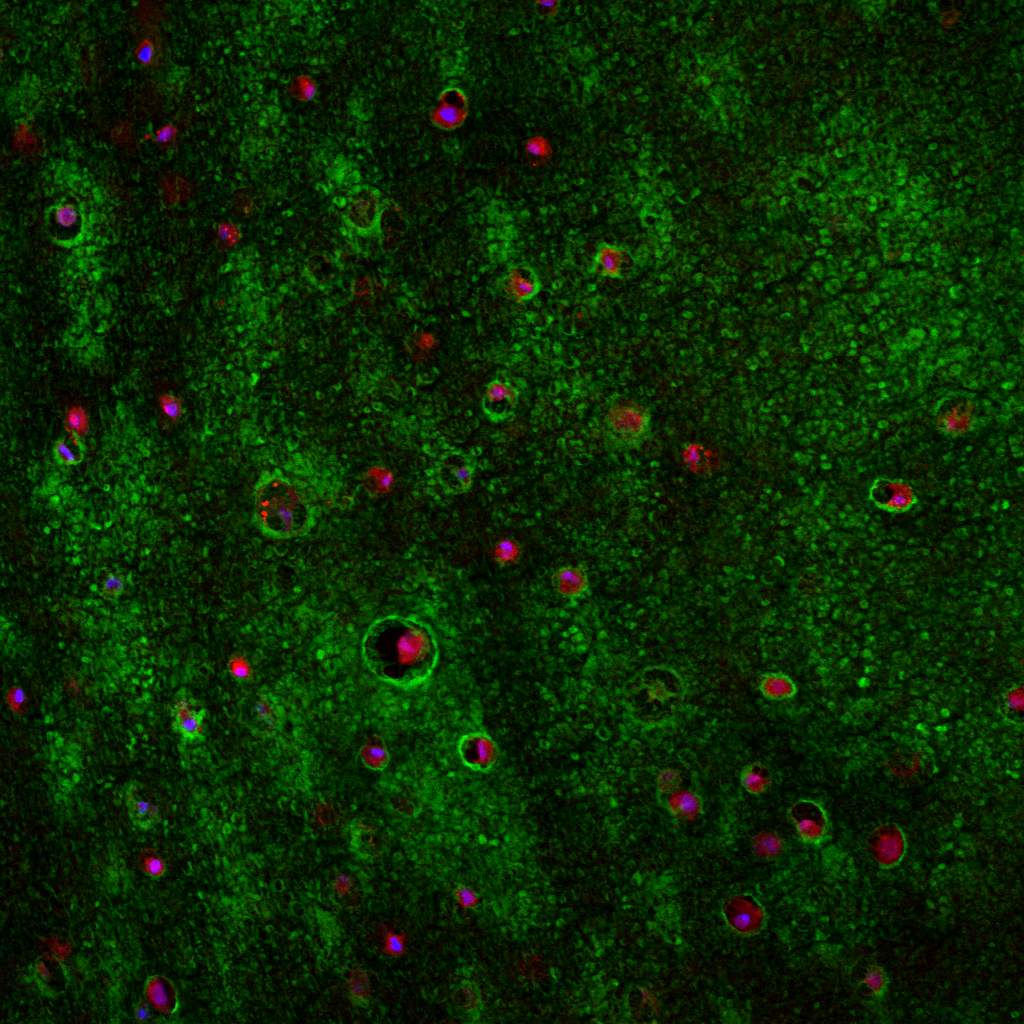
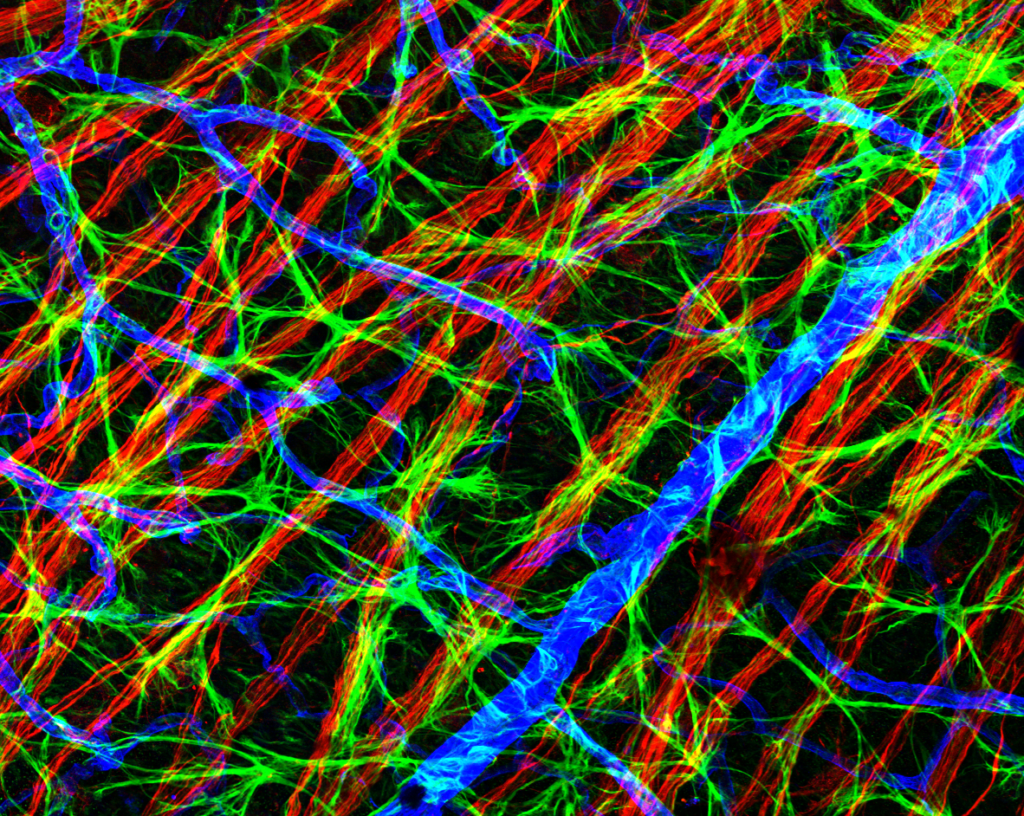
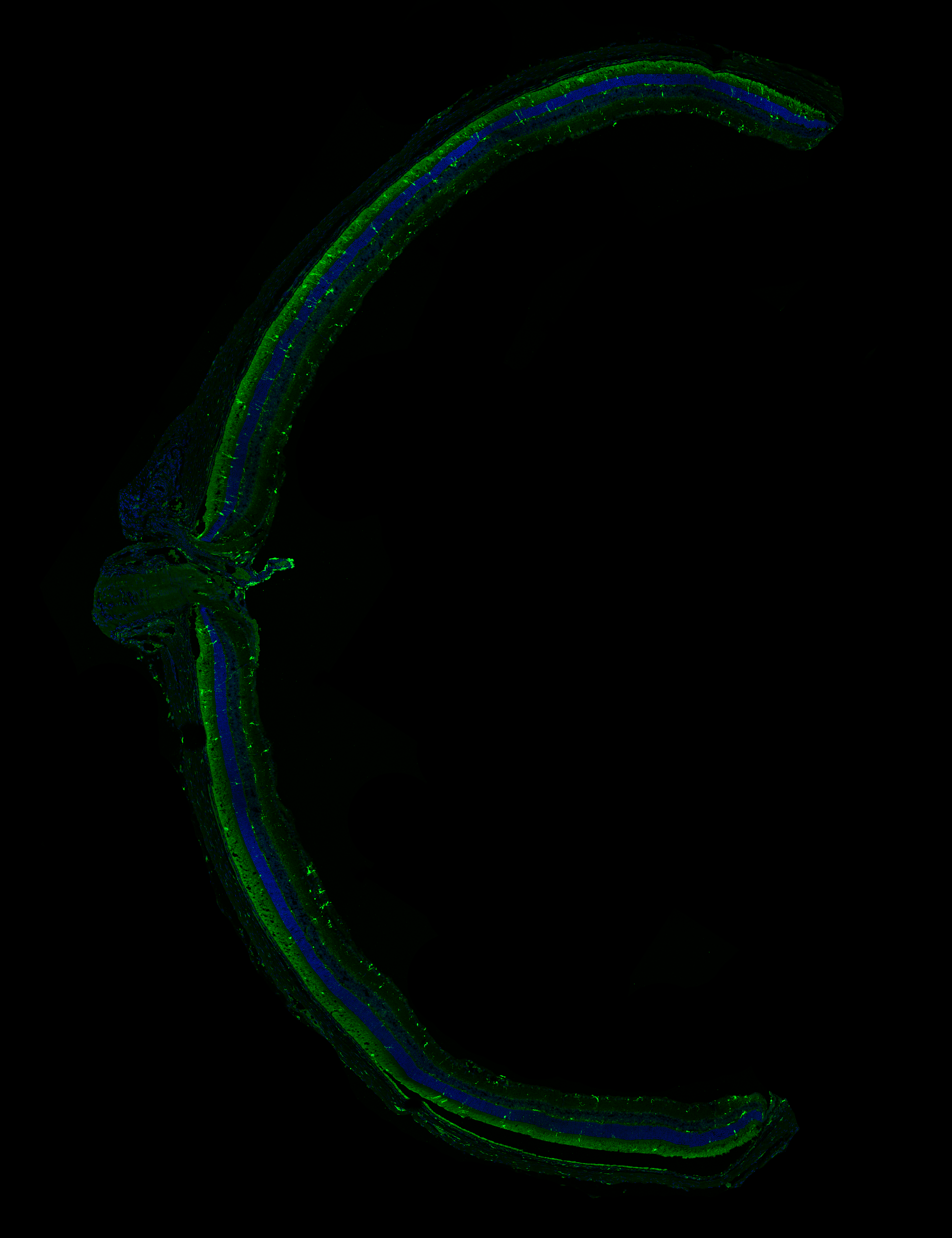
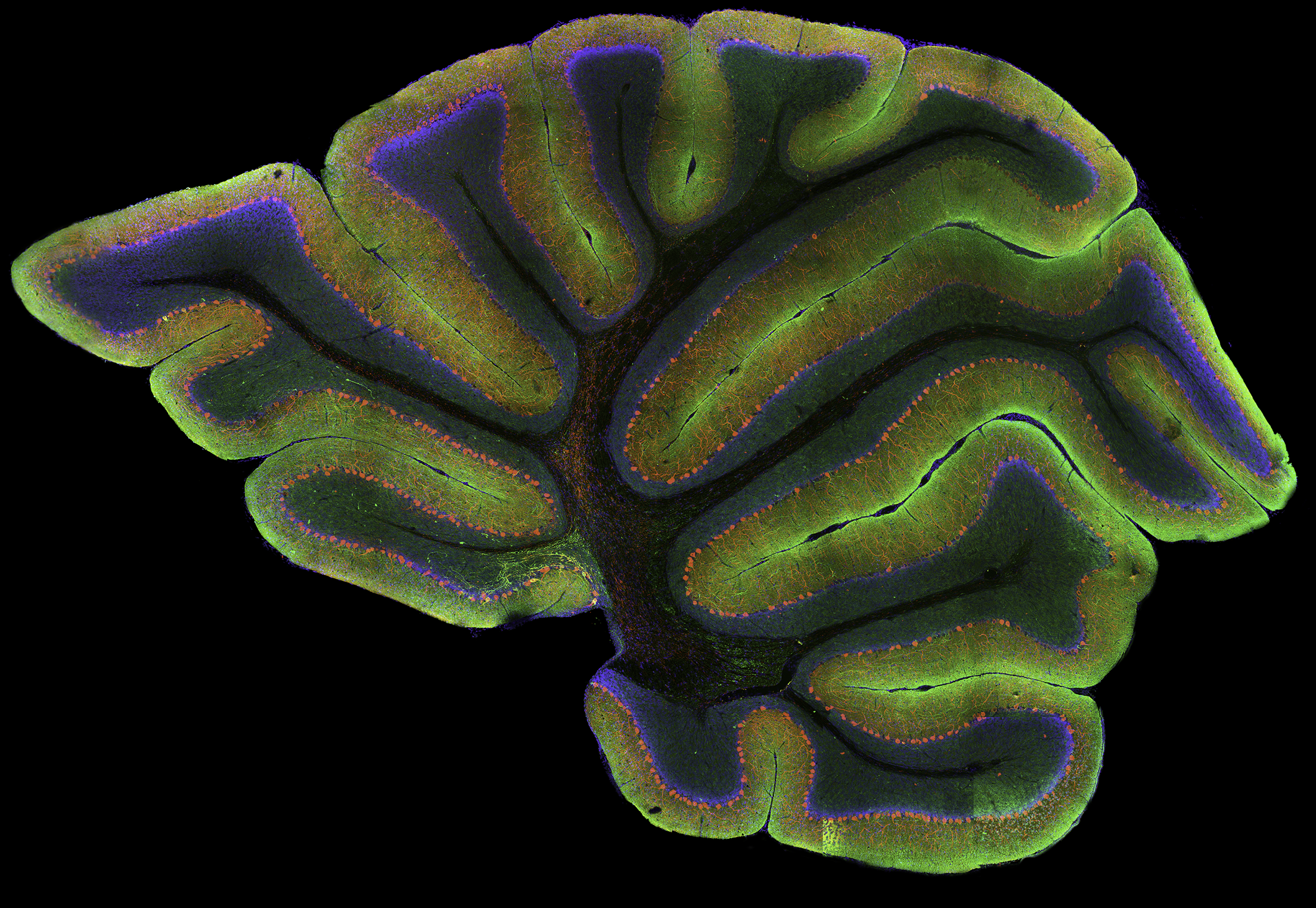 High-resolution, wide-field mosaic of the cerebellum.
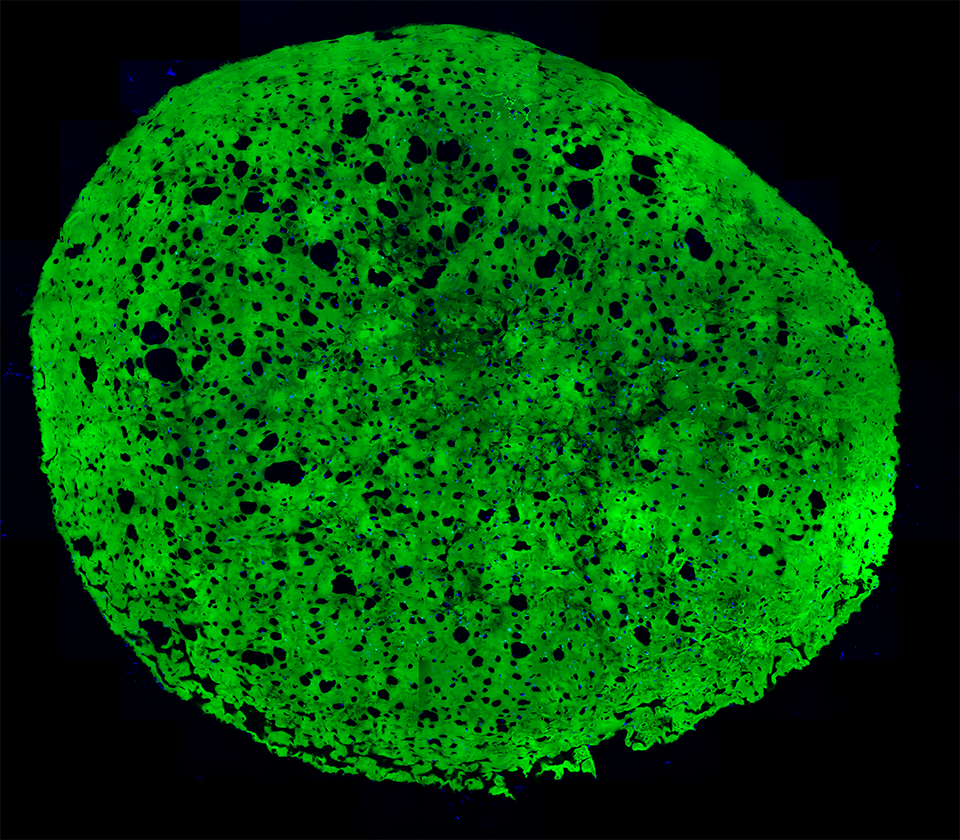
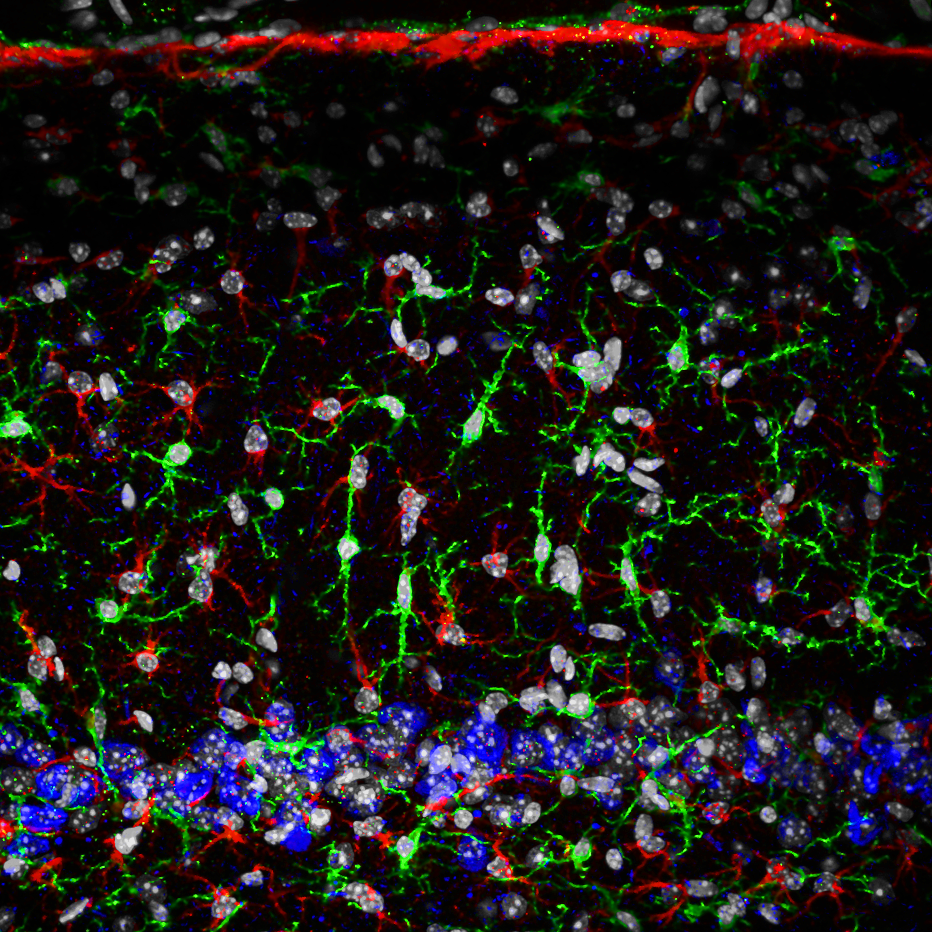
High-resolution, wide-field mosaic of the cerebellum.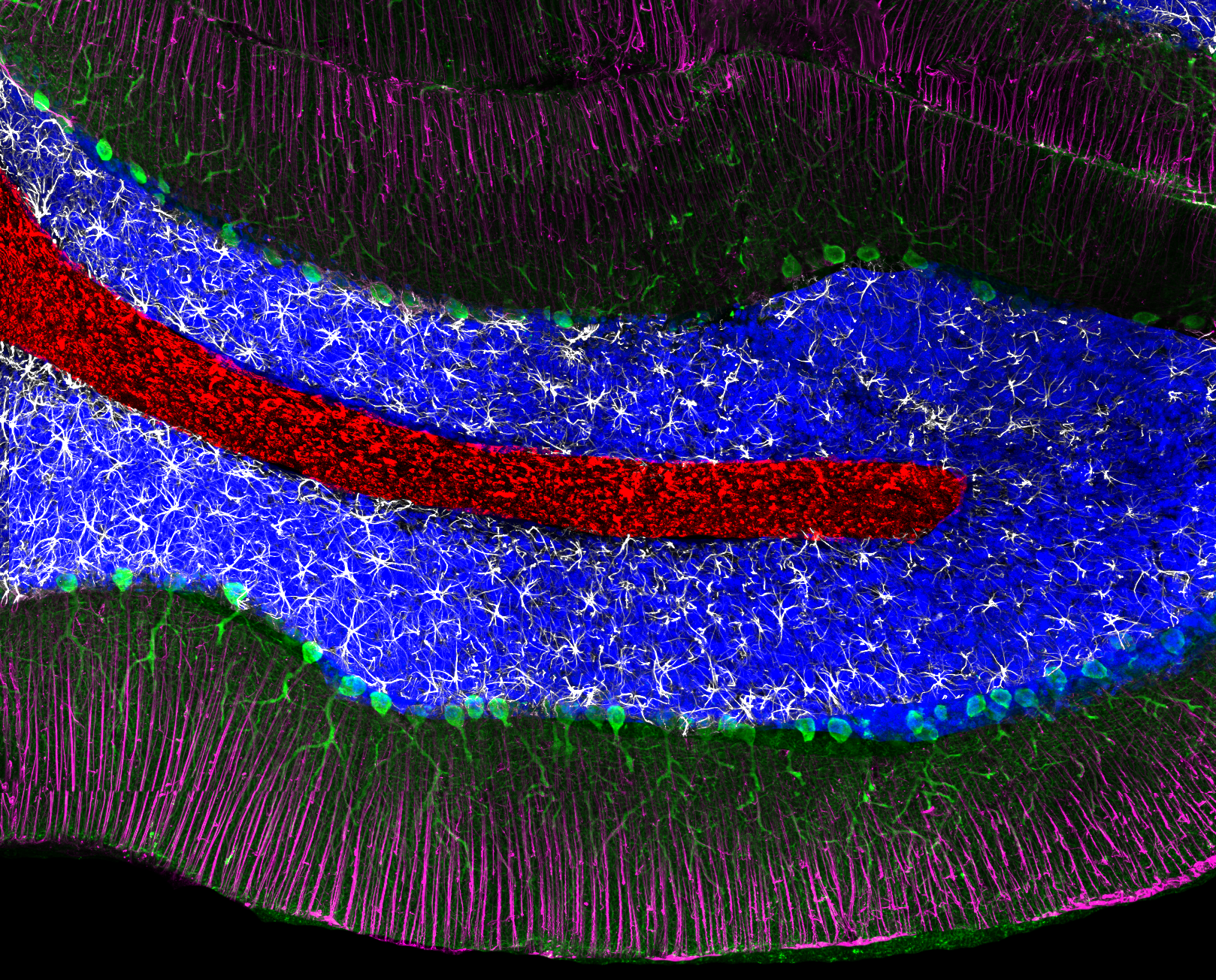 Example of a single tile that comprise the above mosaic.
Example of a single tile that comprise the above mosaic. Olympus flouview 1000
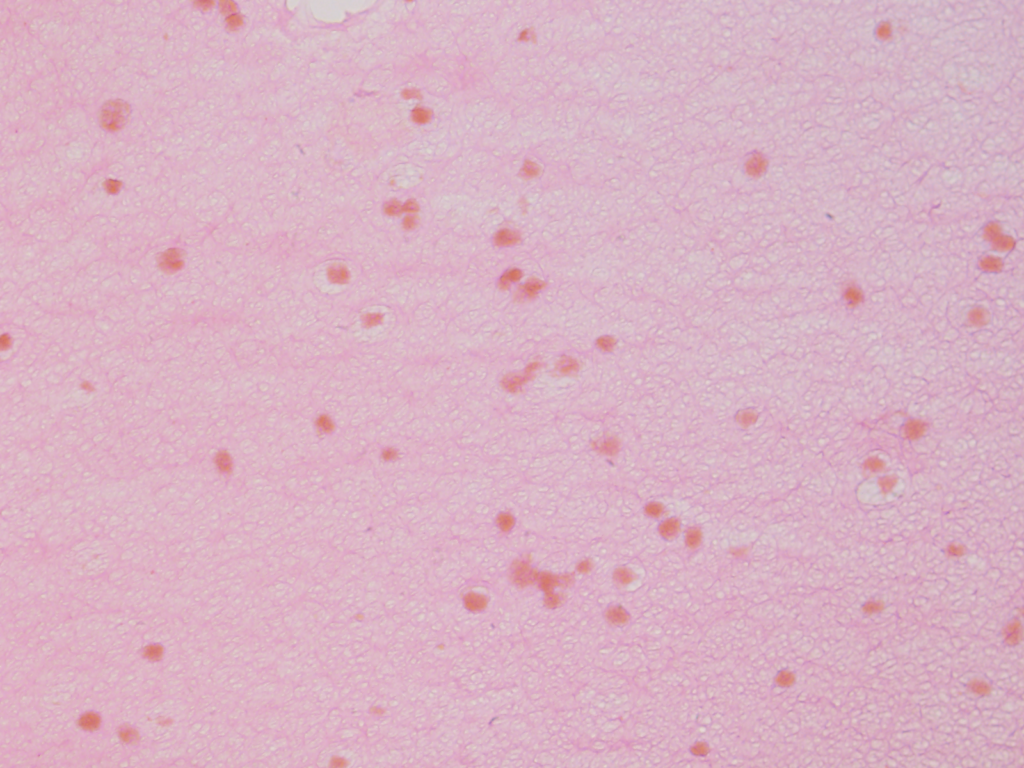
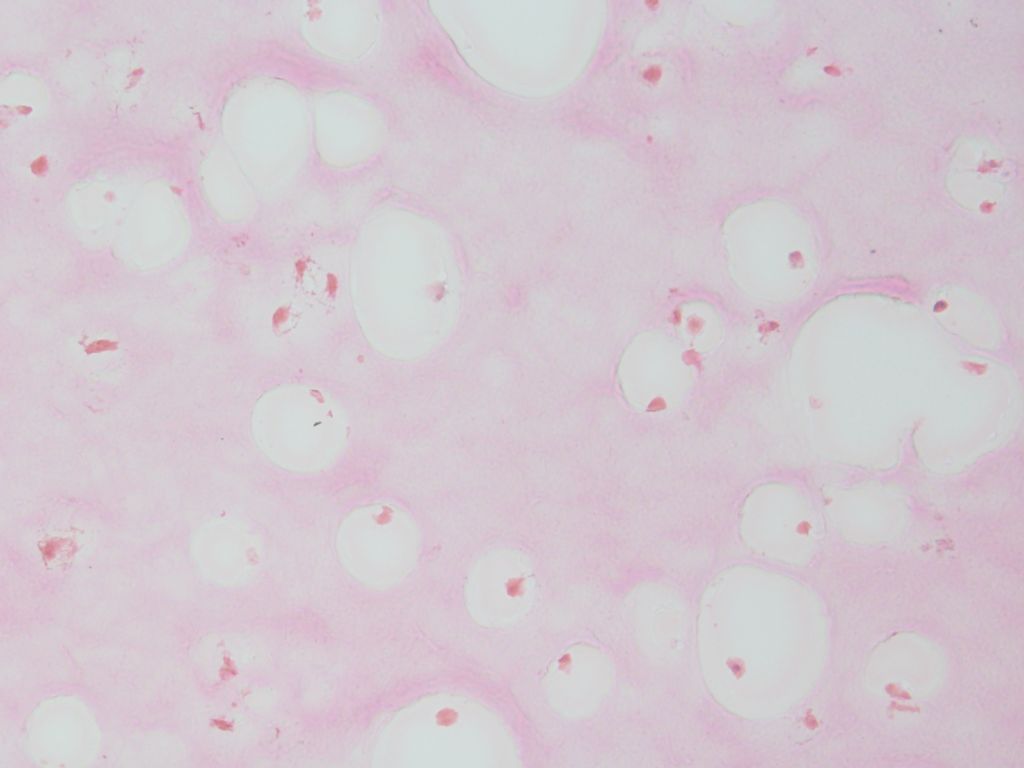
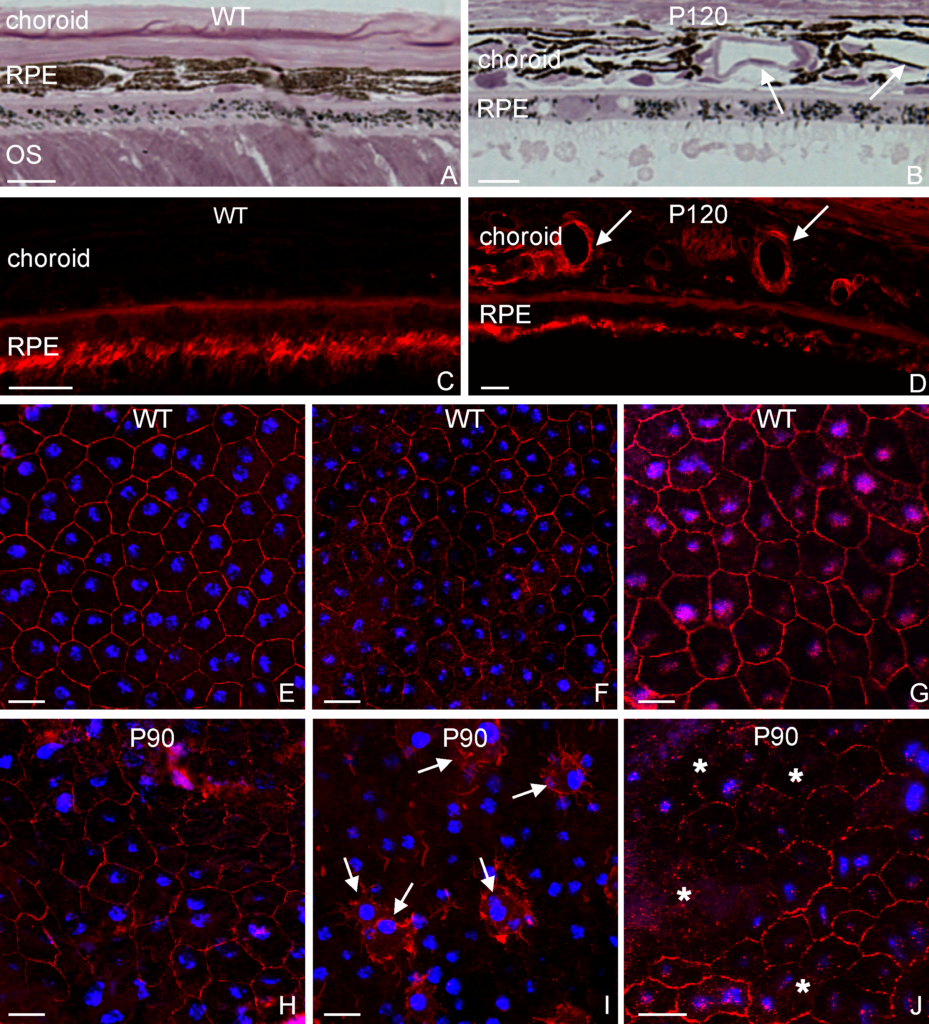
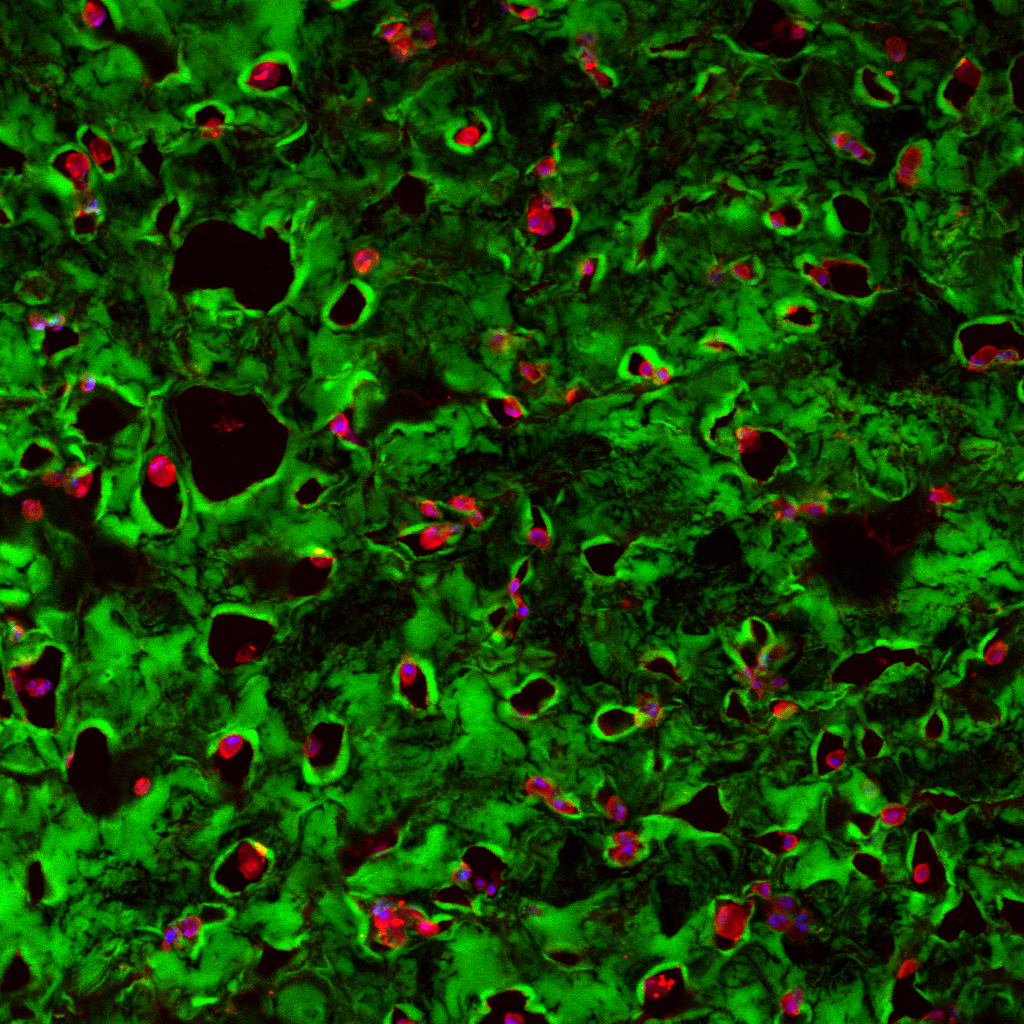
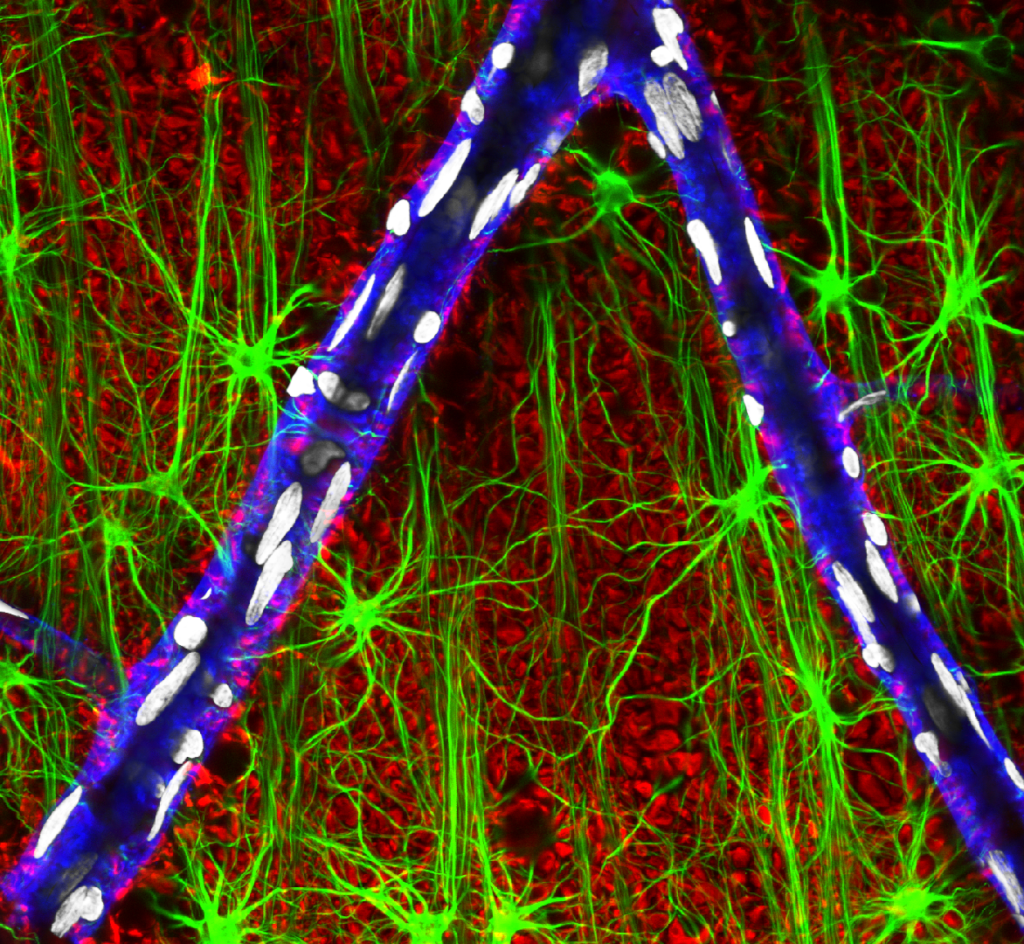
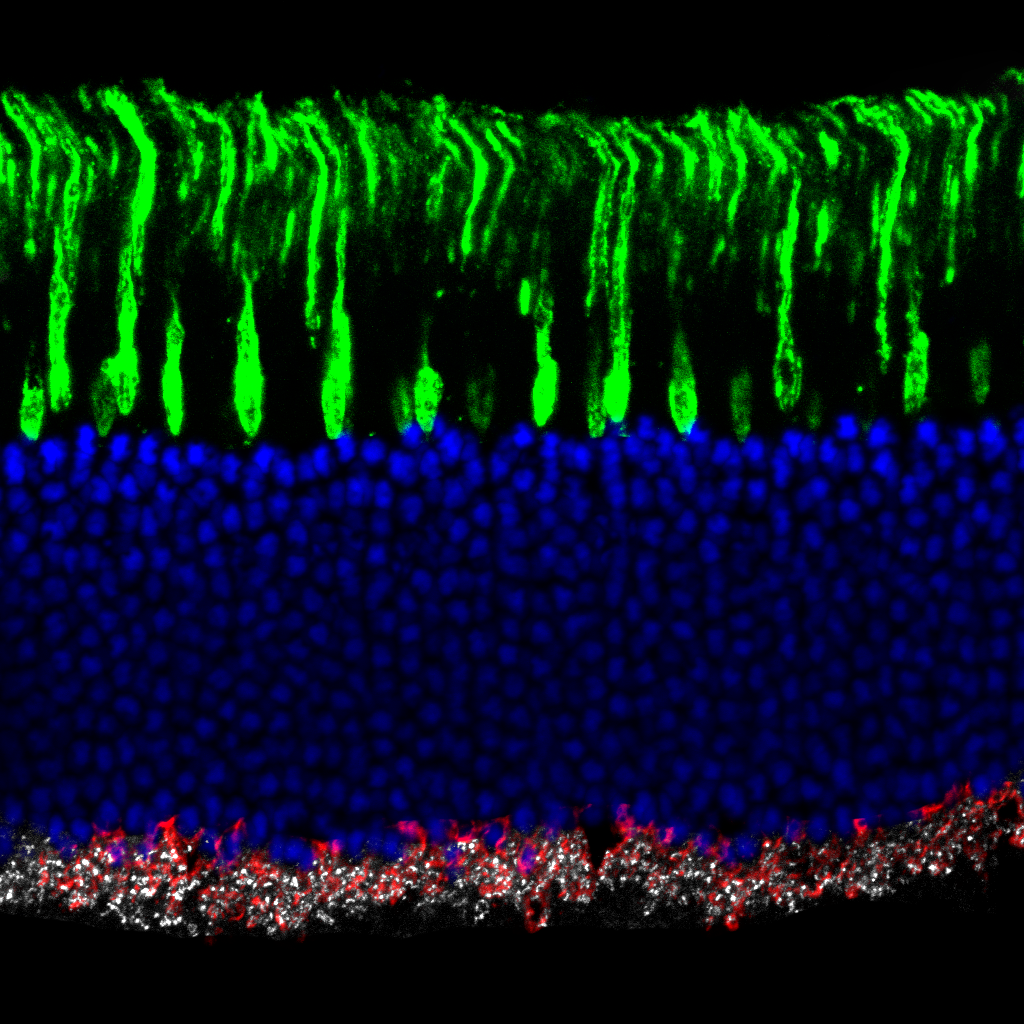
Olympus flouview 1000 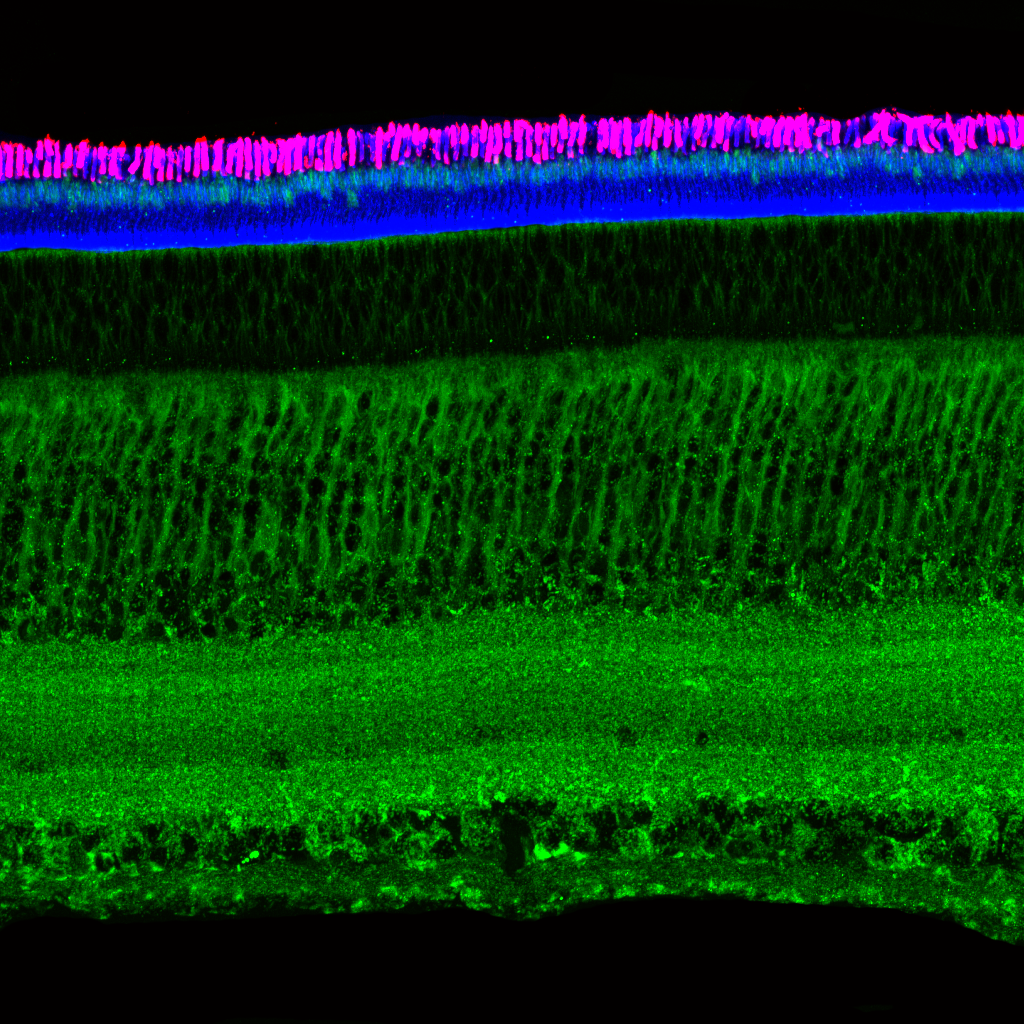 Light micrograph using immunocytochemistry to visualize the cone photoreceptors of the neural retina, magnified 400 times.
Light micrograph using immunocytochemistry to visualize the cone photoreceptors of the neural retina, magnified 400 times.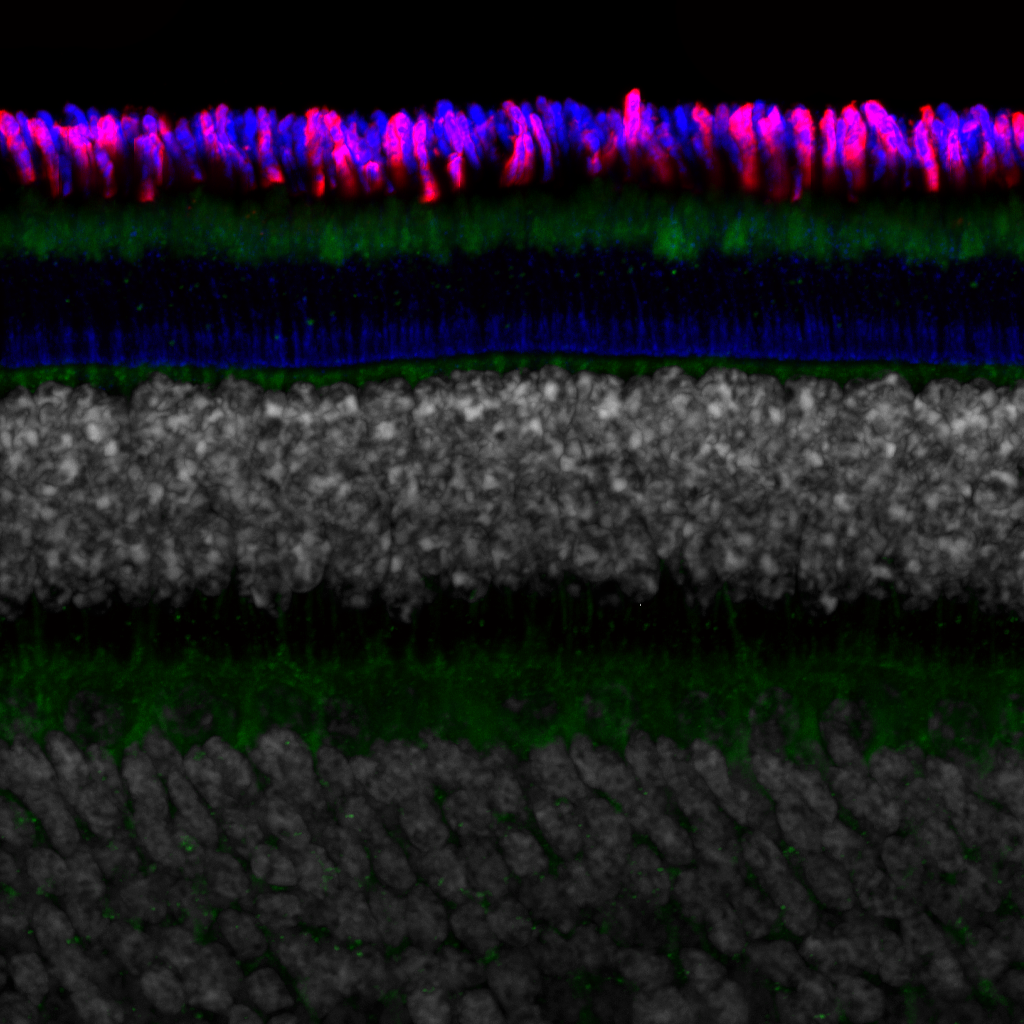
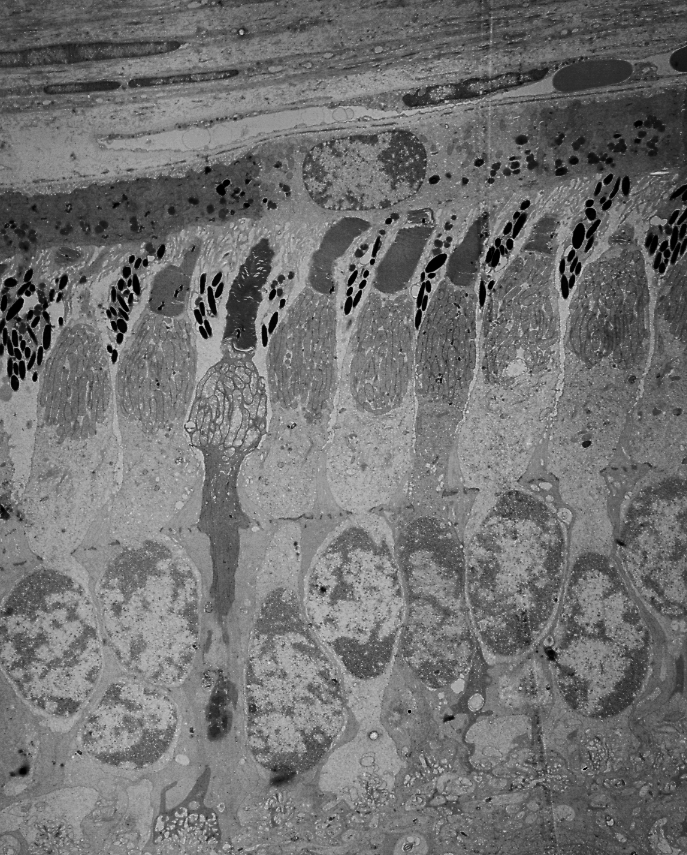
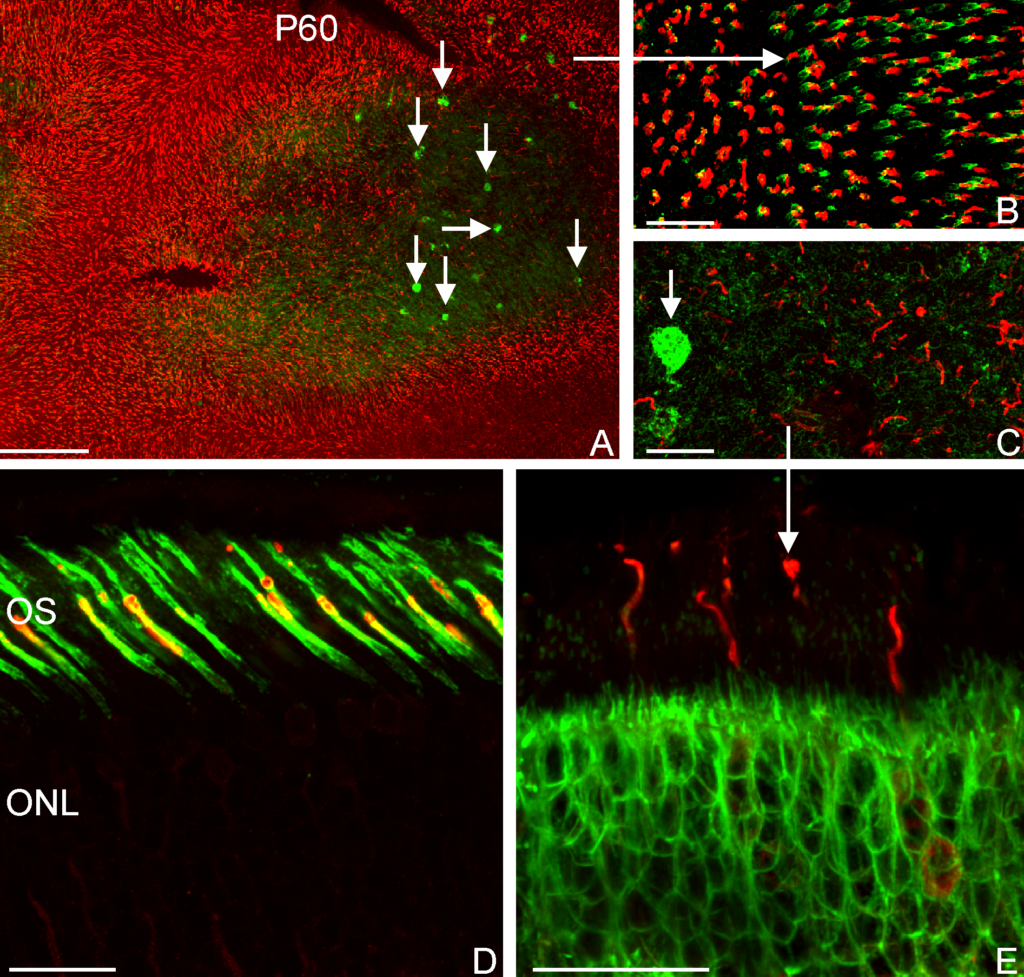
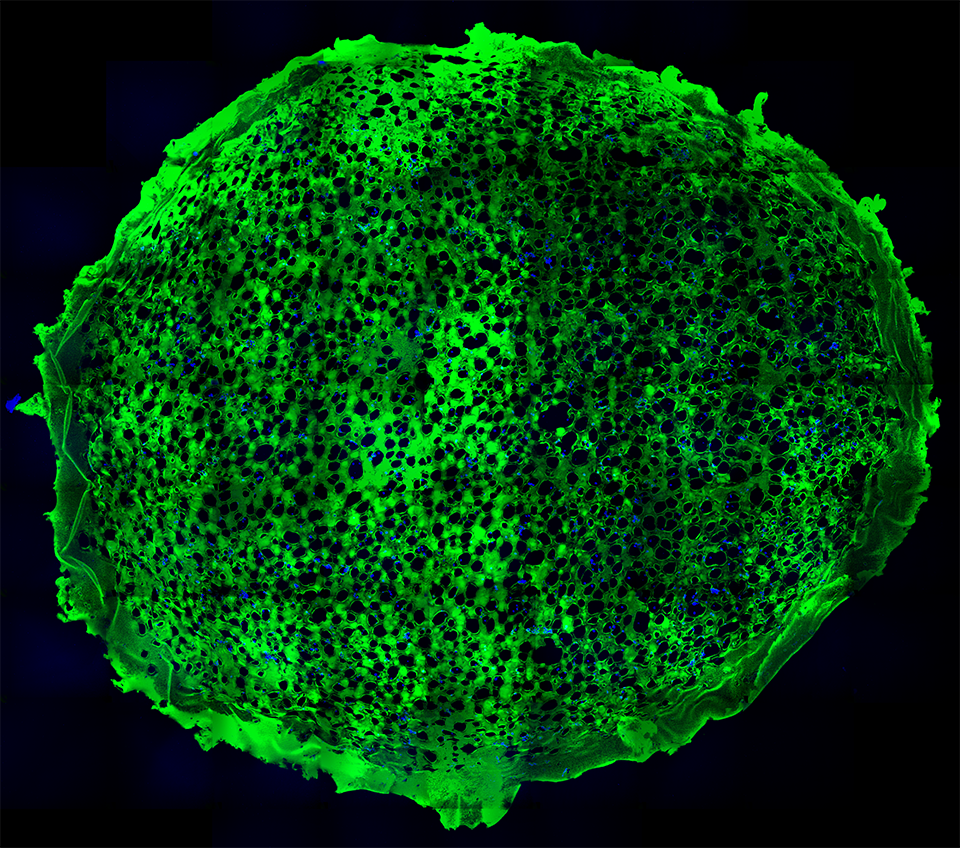
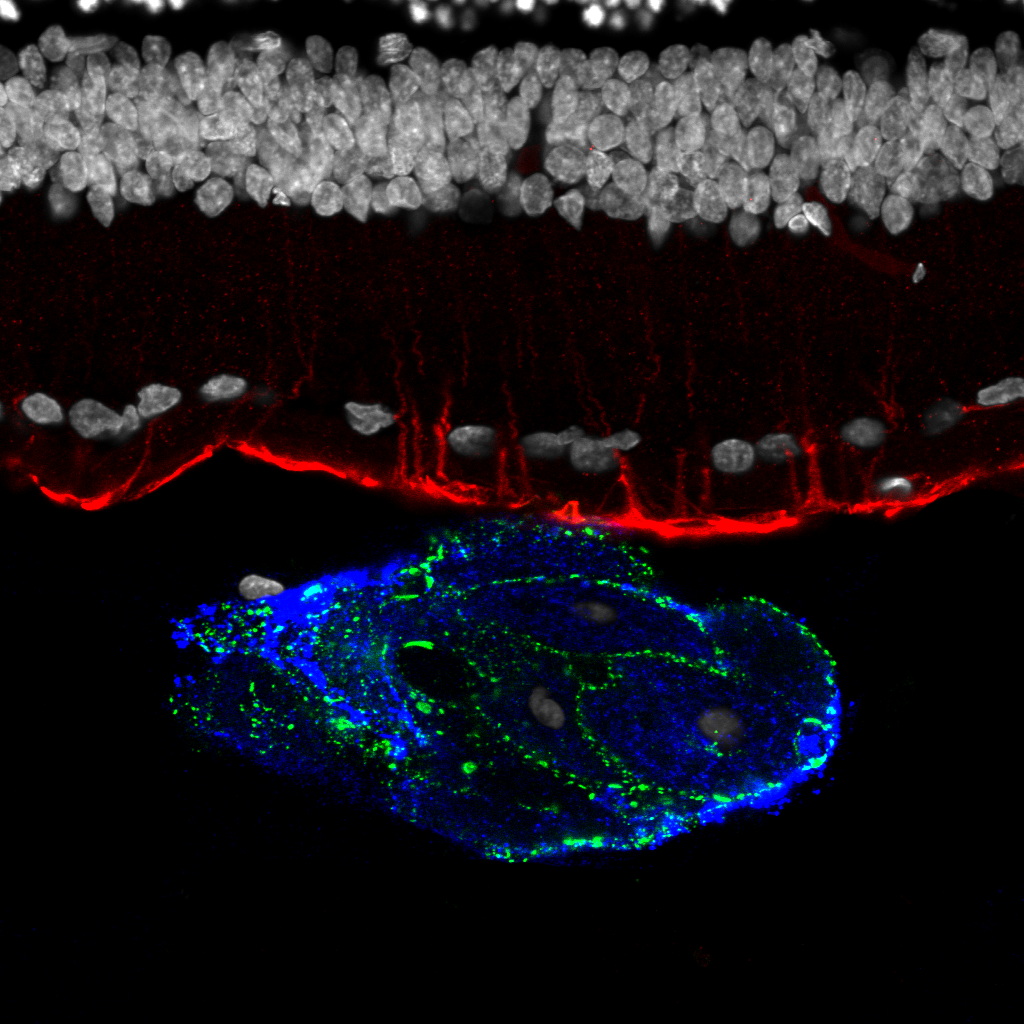
 Electron micrograph of a photoreceptor outer segment, magnified 20,000 times.
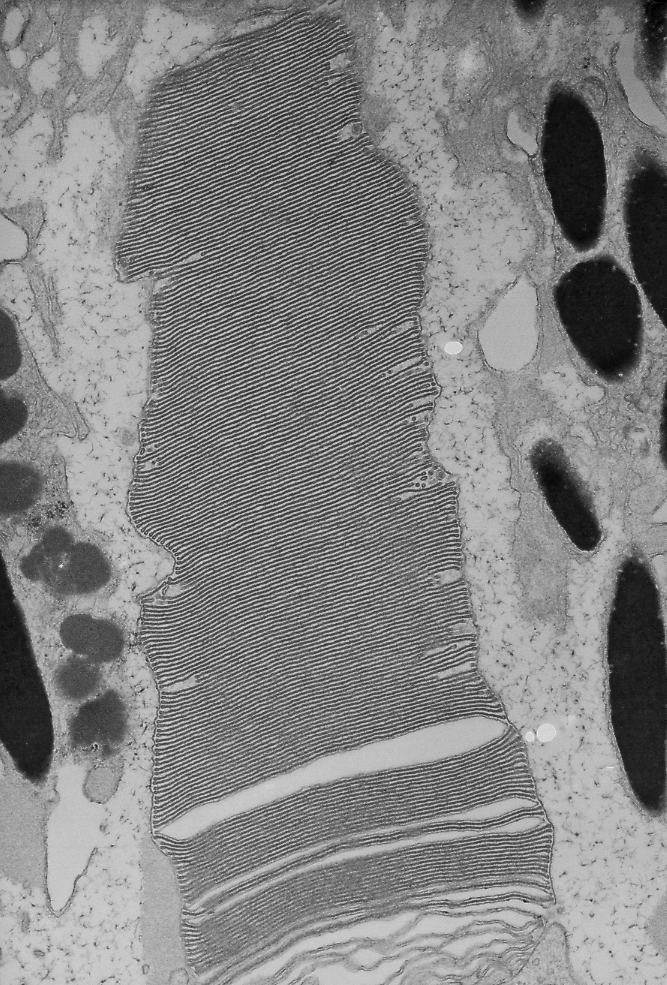
Electron micrograph of a photoreceptor outer segment, magnified 20,000 times.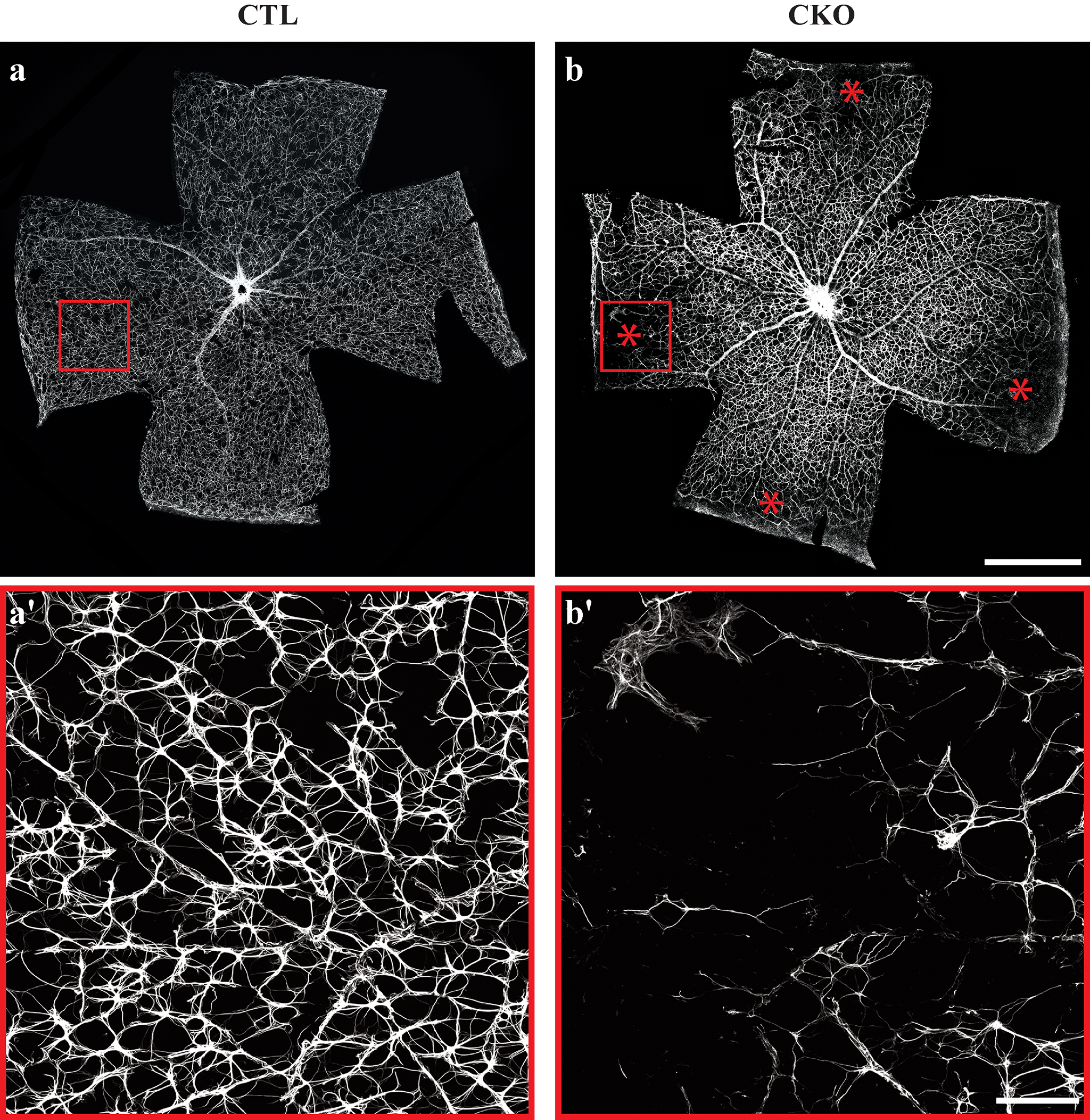
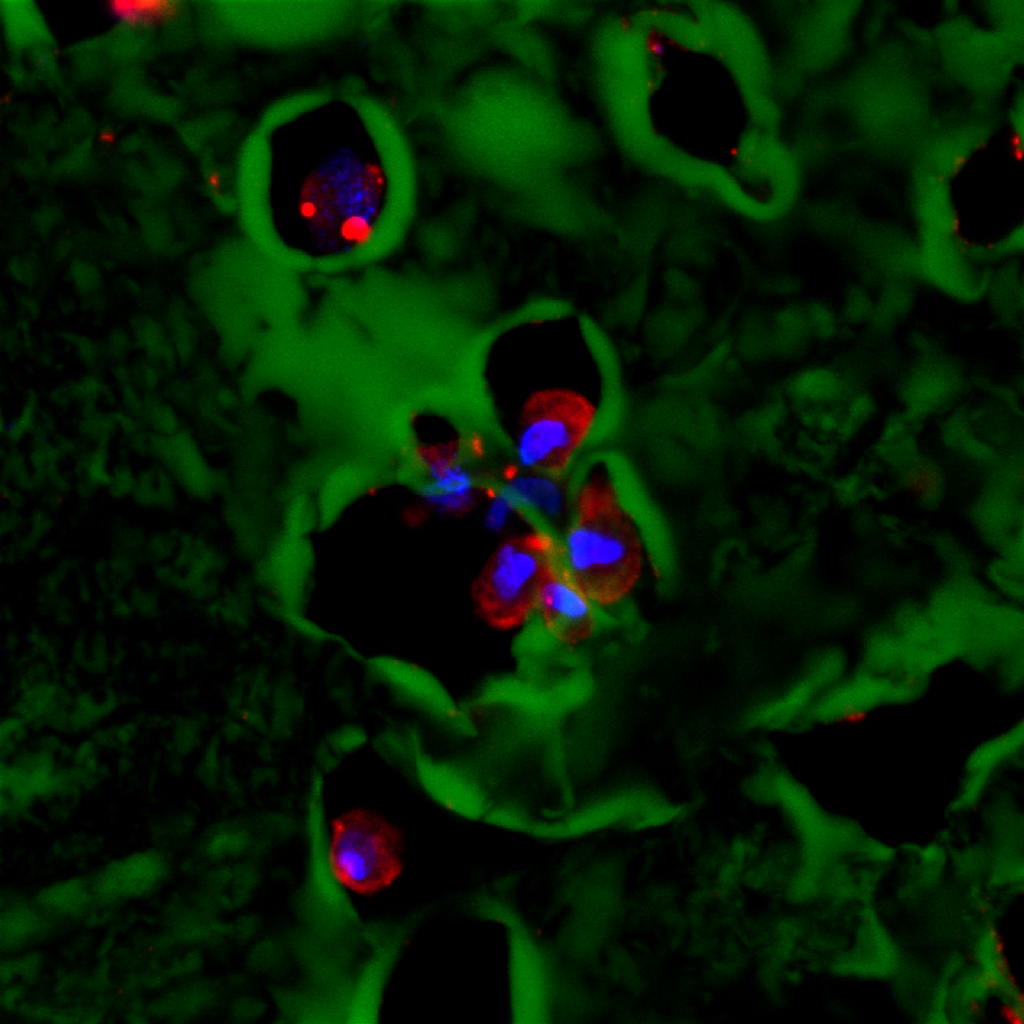
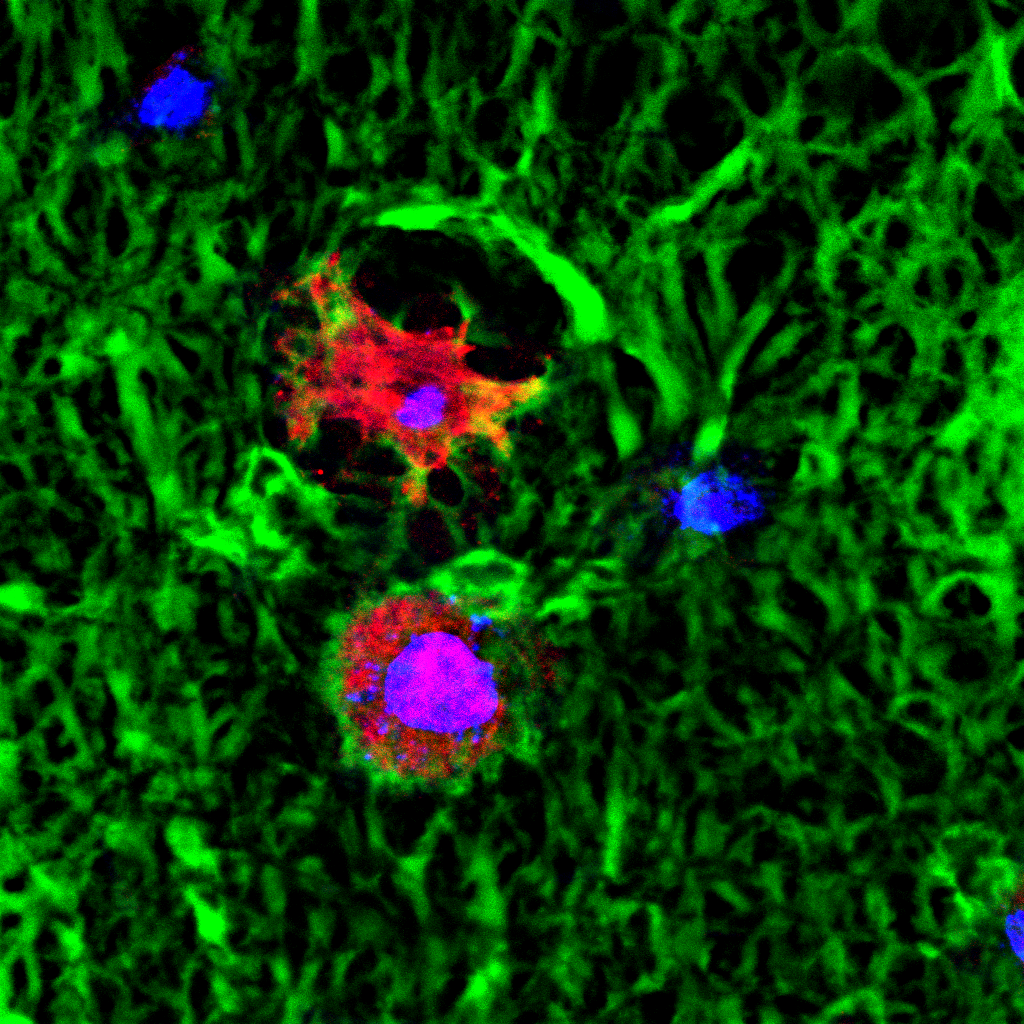
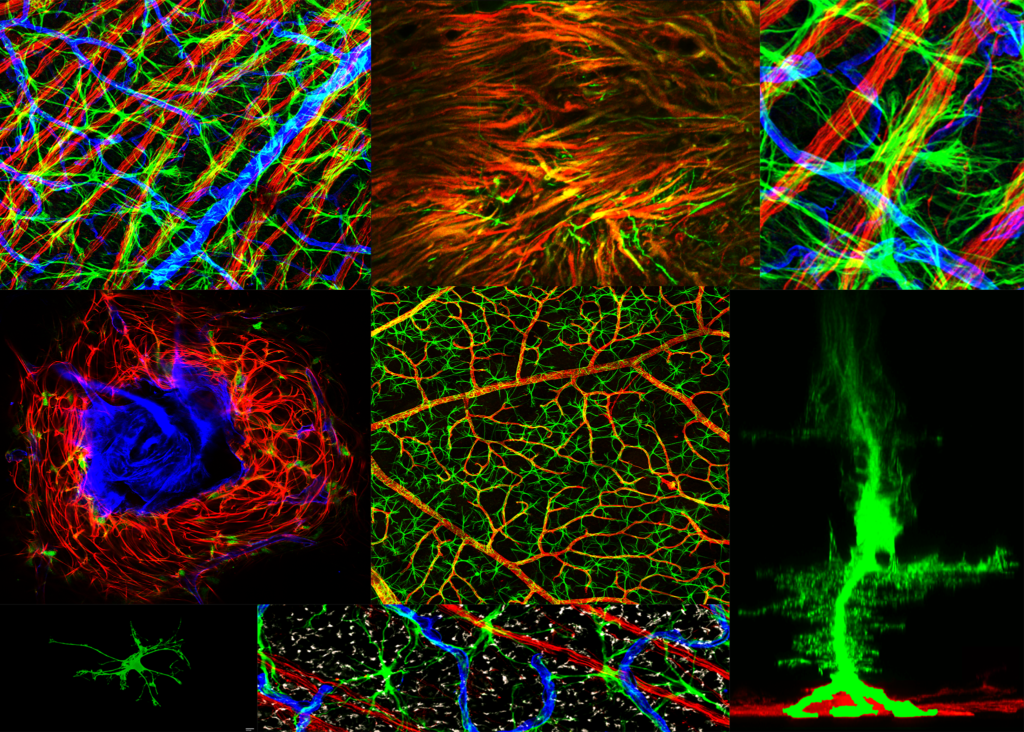
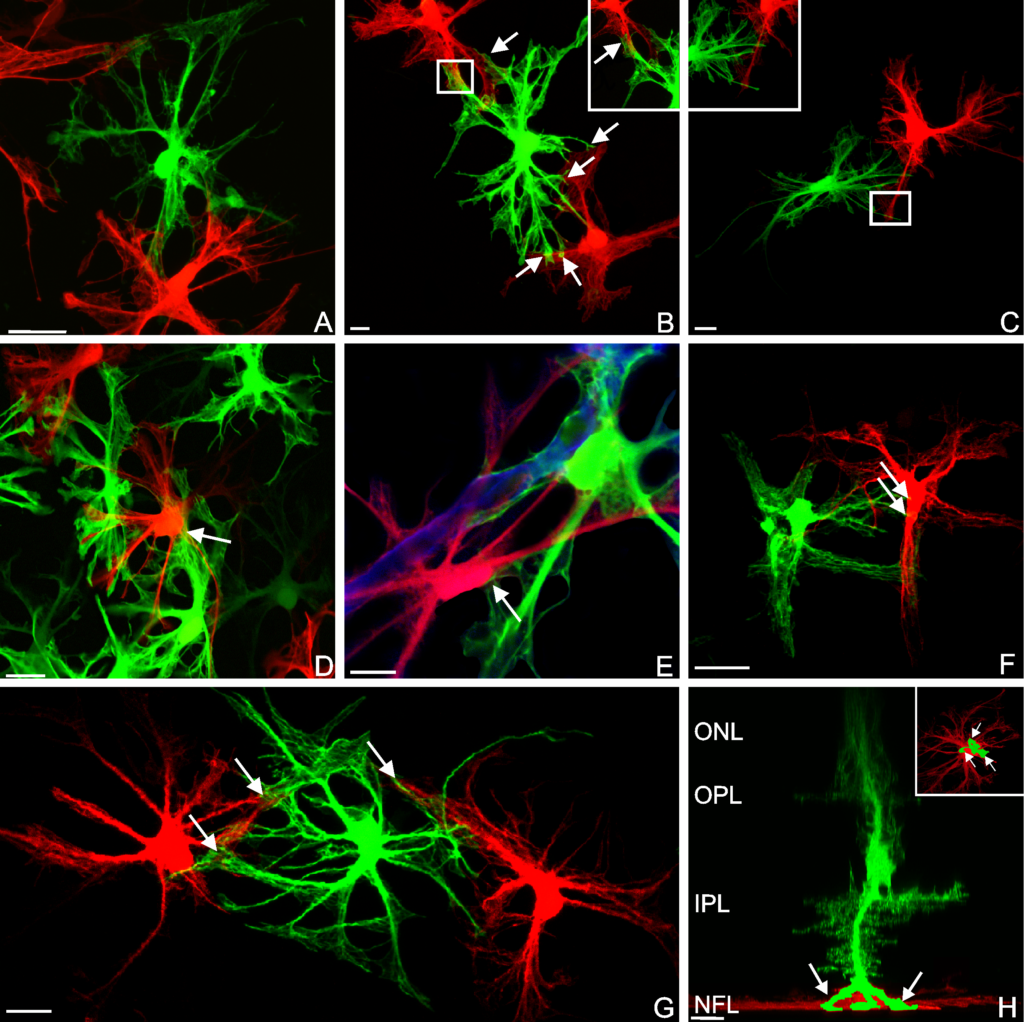
 Composite image.
Composite image.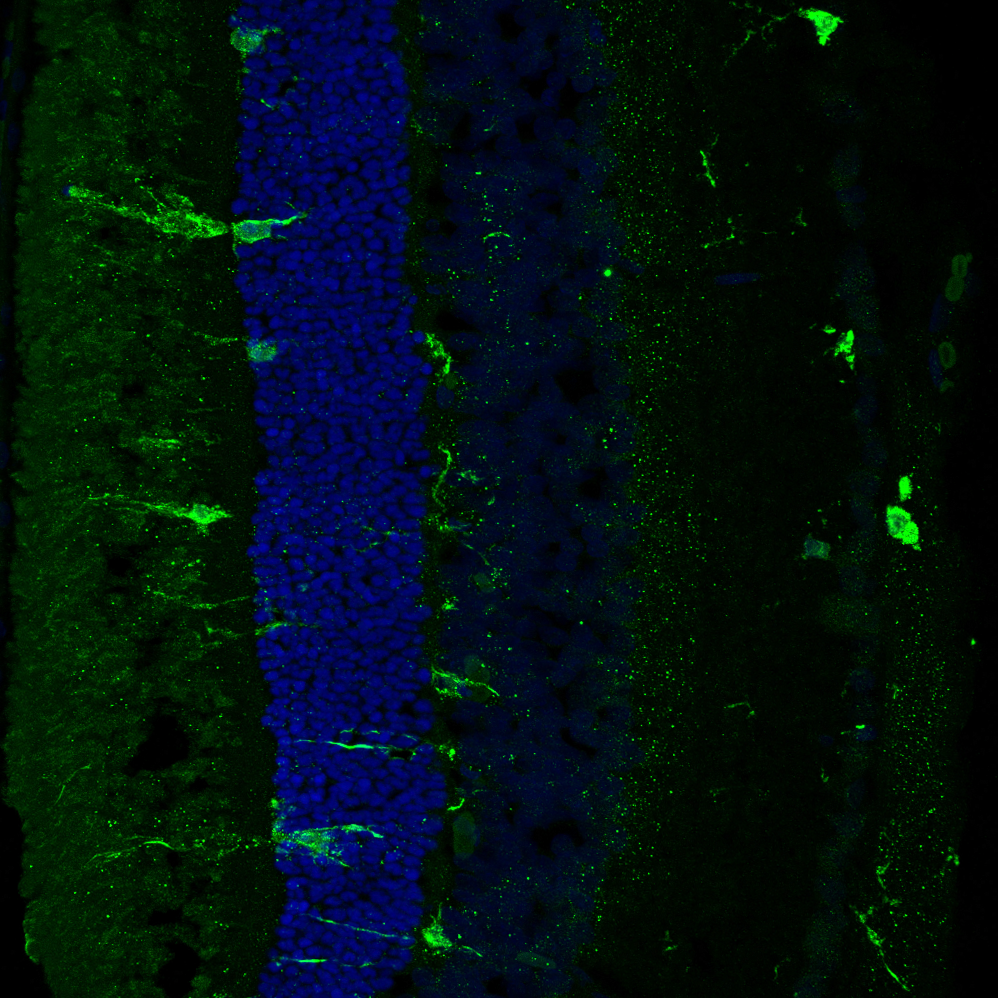 Magnified 400 times.
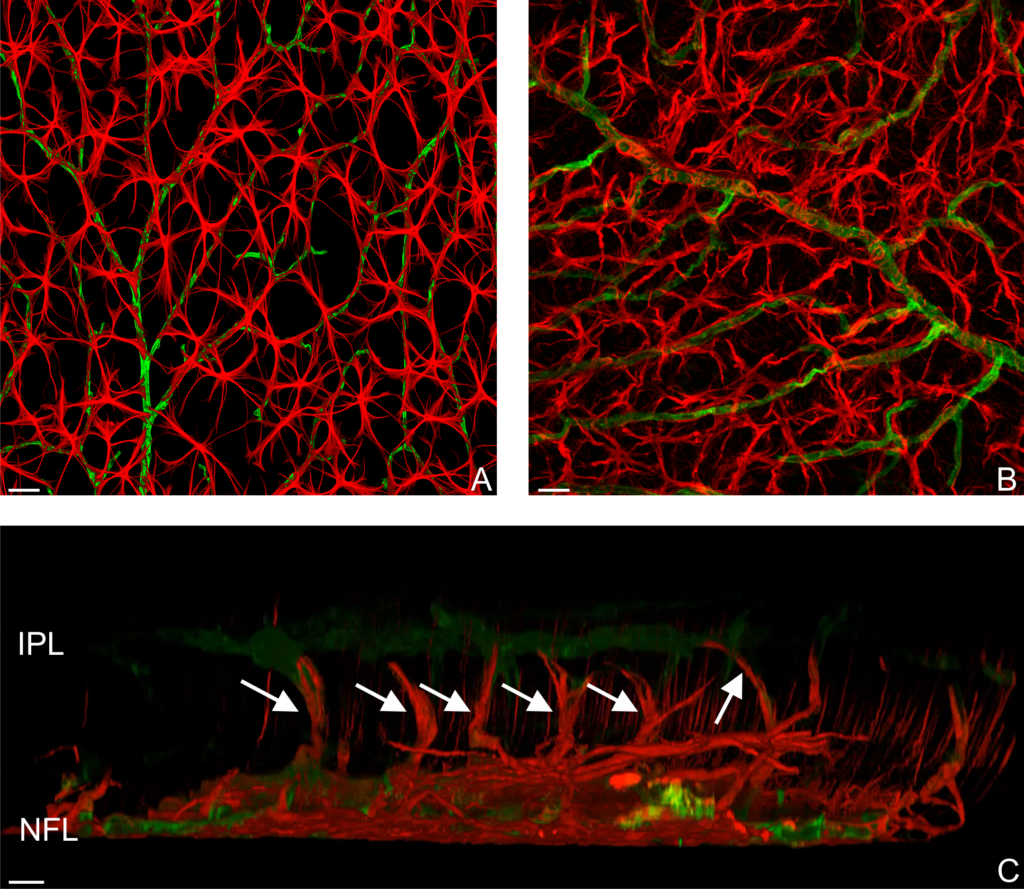
Magnified 400 times. Magnified 600 times.
Magnified 600 times.